Research finds protein regulation linked to cells' growth cycle
By tracking the movement of proteins in a living cell with nanometer-scale precision, Cornell researchers have gained a new insight into the way cells regulate the expression of their genes.
The DNA in every living cell contains blueprints – we call them "genes" – for every protein the cell may need to make. When a particular protein is needed, a regulator protein binds to the DNA chain at the appropriate place, causing the adjacent gene to be "expressed" to make the protein.
But there must also be a mechanism to turn off the gene when enough of the protein has been produced. The cell can't afford to waste energy making proteins it no longer needs.
"Binding of a regulator has been extensively studied, but little was known about how unbinding can be regulated," said Peng Chen, the Peter Debye Professor of Chemistry in the Department of Chemistry and Chemical Biology.
By tagging proteins with fluorescent dyes, Chen and colleagues observed the movements of the proteins down to a resolution of a few nanometers. In a solution of DNA and regulator proteins they saw that when the concentration of the protein rises, another regulator causes the unbinding of the first regulator, stopping expression of the gene.
Now Chen's research group has repeated the experiment in living cells, confirming their original results and adding a surprising new discovery: While unbinding is still tied to the concentration of the protein, it is also linked to the cell's cycle of growth and division. They report their results in the July 6 issue of the journal Nature Communications.
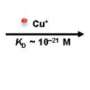
They worked with two proteins known as CueR and ZntR, which regulate the production of proteins that protect bacteria from poisoning by copper and zinc, respectively. Several defensive proteins detoxify the metals or bind to metal atoms and push them out of the cell. The regulator proteins also bind to metal atoms, and when they do they change shape in a way that causes them to bind to DNA at operator sites that turn on the genes to make more defensive proteins. When the intruding metal atoms have been disposed of the cell will be filled with an excess of the "unmetalized" regulator protein, which helps the first regulator to unbind and replaces it on the operator site in a different way that turns off the gene.
Chen learned this by fusing a fluorescent molecule to the proteins in order to observe their actions under a microscope. While a light microscope cannot resolve a single protein molecule it can follow the flashes of light to track one.
To see if what worked in a test tube was also true in a living cell, the researchers placed E-coli bacteria with tagged regulator proteins in a nutrient solution under a cover glass on a microscope slide. Zapping the slide every 60 milliseconds with a laser to induce fluorescence, they created a time-lapse movie of protein movements.
The experiment confirmed the earlier finding that unbinding the first regulator and halting gene transcription followed an increase in the concentration of the unmetalized regulator protein. But there was also a surprise: the unbinding events were spread over a larger area, showing that the chromosome was "less condensed." Ordinarily, DNA is tightly coiled and "condensed" in the chromosome, but it becomes less condensed when the cell grows so that the chromosome can be duplicated, and again becomes tightly condensed when the duplication finishes and the cell is ready to divide. So the regulator unbinding, and the associated gene expression regulation, is tied to the growth cycle of the cell.
"It was a very surprising observation," Chen said. "The protein has two forms, bound [to a metal atom] and unbound. The different forms respond to chromosome condensation in different ways." Although the research might lead to new ways to kill harmful bacteria, "How it relates to antibiotics I can only speculate," Chen said. "I'm excited about how the fundamental biology works."
Journal information: Nature Communications
Provided by Cornell University