July 21, 2015 feature
Macroscopic quantum phenomena discovered in ice
(Phys.org)—Scientists have discovered an anomaly in the properties of ice at very cold temperatures near 20 K, which they believe can be explained by the quantum tunneling of multiple protons simultaneously. The finding is a rare instance of quantum phenomena emerging on the macroscopic scale, and is even more unusual because it is only the second time—the first being superconductivity—that macroscopic quantum phenomena have been observed in a system that is based on fermions, which include protons, electrons, and all other matter particles. Other systems exhibiting macroscopic quantum phenomena have been based on photons, a type of boson, which mediate the forces between matter.
The scientists, Fei Yen at the Chinese Academy of Sciences and Tian Gao at Shanghai University of Electric Power, have published a paper on the anomaly in ice in a recent issue of The Journal of Physical Chemistry Letters.
Ice rules
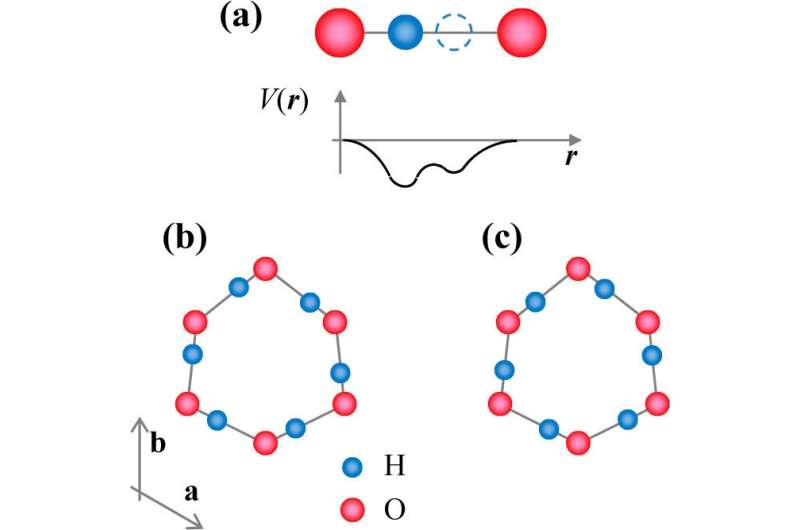
As the scientists explain, when water freezes, the oxygen atoms in the ice become ordered into a puckered hexagonal-like lattice. The hydrogen atoms, on the other hand, remain quite disordered. This freezing process is governed by the two "ice rules," which state that only one hydrogen atom can reside between two oxygen atoms, while each oxygen atom can be bonded to four hydrogen atoms by bonds of two different lengths.
Because of the two different bond lengths, between each pair of oxygen atoms, there are two sites available for a single hydrogen atom (which has lost its electron and simply becomes a proton). At high enough temperatures (above 136 K), the protons have enough energy to move between the two sites. However, when the temperature falls below 136 K, the protons no longer have enough energy to move between sites and randomly "freeze" in one of the sites.
Tunneling protons
Now in the new study, the scientists have found that the protons actually can move between these two sites even at very cold temperatures of less than 20 K. The protons cannot move by classical means (as they don't have enough energy), but by quantum tunneling through the classical energy barrier.
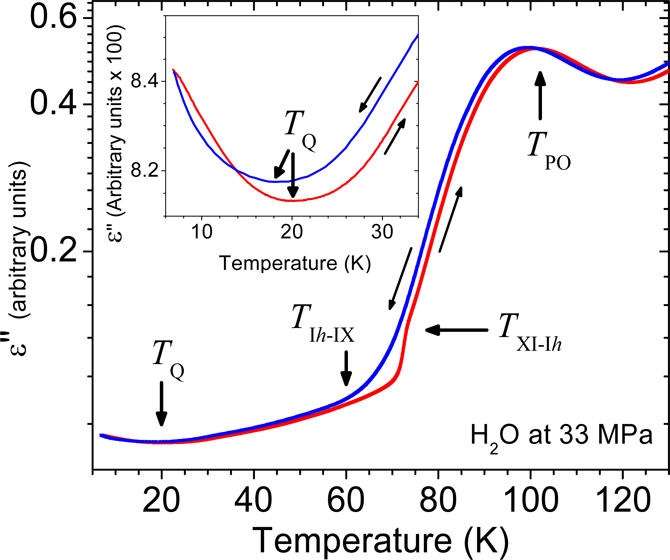
Although quantum tunneling occurs due to the wave-like nature of particles at the quantum scale, and is not possible at the macroscopic level, here the scientists demonstrate that the total combined effects of quantum tunneling can be witnessed and measured at the macroscopic level. The macroscopic evidence for the quantum phenomena comes from measuring the dielectric properties of ice. As a dielectric material, ice is an electrical insulator, but in the presence of an electric field the molecules become polarized so that they align themselves with the electric field.
To investigate the dielectric properties of ice, the scientists made measurements on a pair of platinum plates inserted into ice inside a Teflon container, all of which was frozen in a cryostat. The researchers discovered the existence of a minimum in the imaginary part of the dielectric constant of ice at 20 K but no change in the real part. As the scientists explain, the physical meaning of this anomaly can be interpreted as an increase in the movement of charges—in other words, protons moving back and forth between sites. They also found that no anomaly occurs in heavy ice (i.e., deuterium, which contains a proton and a neutron), indicating that the discovery also exhibits an isotope effect.
How do the protons do it?
To better understand what may be happening at the atomic level, the researchers again turned to the ice rules. According to these rules, single protons cannot move between sites one at a time, as this interferes with the highly ordered crystal structure of ice. However, the ice rules remain preserved if all six protons within a hexagonal ring move at the same time, which suggests that the six protons engage in correlated tunneling.
But correlated tunneling poses another problem. According to the laws of quantum mechanics, for several protons to tunnel simultaneously, they must all have the same wave function and occupy the same ground state. However, this arrangement directly violates the Pauli exclusion principle, which expressly states that no two identical fermions can simultaneously occupy the same quantum state.
To overcome this problem, the scientists conjecture that the protons tunnel in pairs because a proton pair can act as a boson, which is allowed to collapse into its ground state because it is not subject to the exclusion principle. This proposal is similar to the underlying mechanism of superconductivity, which is caused by paired electrons forming a "Cooper pair" that also acts as a boson and condenses into its ground state. In both situations, pairing allows for multiple fermions to move together simultaneously, giving rise to macroscopic quantum phenomena that wouldn't otherwise be allowed.
Going forward
Observing macroscopic quantum phenomena is not only of fundamental interest, but the researchers predict that it may be very useful one day, as well.
"With new phenomena usually comes new applications, and there is no exception in the realm of macroscopic quantum phenomena; see, for instance, what has been accomplished with superconductivity and lasers," Yen told Phys.org. "As for correlated proton tunneling, perhaps a higher accuracy can be obtained on some of the fundamental constants, such as the case of the quantum hall effect, where the von Klitzing and fine structure constants have been determined to higher precision."
In the future, the scientists plan to look for the same quantum phenomenon in related systems.
"We believe that correlated proton tunneling on the macroscopic scale is not strictly limited to ice and should also occur in other hydrogen-based compounds provided the temperature is low enough," Yen said. "Currently, we are still investigating water ice, though at higher pressures, where the ices crystallize in tetragonal, monoclinic or interpenetrating cubic lattices to see if the same phenomenon is also present."
More information: Fei Yen and Tian Gao. "Dielectric Anomaly in Ice near 20 K: Evidence of Macroscopic Quantum Phenomena." The Journal of Physical Chemistry Letters. DOI: 10.1021/acs.jpclett.5b00797
Journal information: Journal of Physical Chemistry Letters
© 2015 Phys.org





















