New microscope technique could speed identification of deadly bacteria

A new way of rapidly identifying bacteria, which requires a slight modification to a simple microscope, may change the way doctors approach treatment for patients who develop potentially deadly infections and may also help the food industry screen against contamination with harmful pathogens, according to researchers at the Korea Advanced Institute of Science and Technology (KAIST) in Daejeon, South Korea.
Described this week in The Optical Society (OSA) journal, Optics Express, the new approach involves bouncing laser light off individual bacteria under the microscope, creating holographic images of them, and then using a mathematical transformation and computer software to analyze the images and identify them by comparing them to other, known bacteria. The software uses a machine-learning algorithm—the sort computers connected to security cameras might use for automated facial recognition.
If the approach proves to be effective in controlled clinical trials, it could lead to a powerful new way of routinely and almost immediately identifying dangerous bacteria at the bedside—much faster than the days it normally takes to grow or "culture" the microbes in the laboratory from a patient's blood, which is still the gold standard in the health care industry for making a definitive diagnosis. Also routinely used today is a newer method for rapidly identifying bacteria based on a DNA-analysis technique called quantitative polymerase chain reaction (qPCR), but it still may take hours to return a result, and it requires expensive sample preparation that is often cost prohibitive for routine use in rural or resource poor settings around the world.
The challenge of meeting clinical needs in the developing world was one of the motivations behind the work, according to the KAIST team. They wanted to find an inexpensive method that could surpass the speed of qPCR.
"Employing laser holographic techniques, we achieved rapid and label-free identification of bacterial species at the single bacterium level with a single-shot measurement," said physicist YongKeun Park, who led the KAIST team. "This means the present method can be utilized as a prescreening test for point-of-care bacterial diagnosis for various applications including medicine and food hygiene."
"We have also developed a compact portable device, so called quantitative phase imaging unit (QPIU) (Optics Letters 39 (12), 3630-3633), to convert an simple existing microscope to a holographic one, in order to measure light scattering patterns of individual bacteria, which can then be used to identify bacteria species for rural areas and developing countries," said Park. "Our team has plans to go to Tanzania next month for a field test."
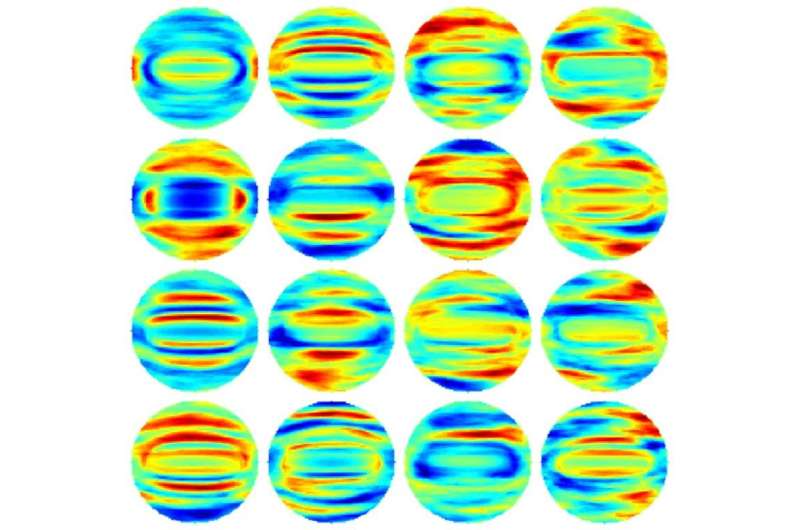
Why Speed Matters in Infection Control
In hospitals and clinics worldwide, bacterial infections are a major source of illness, often slowing recovery or making matters much worse for people who are admitted to inpatient care for routine or emergency treatment. In the most severe cases, bacterial poisoning causes severe disease and syndromes like sepsis, meningitis, pneumonia, and gastroenteritis—all of which can be deadly unless the patient is given immediate and appropriate treatment.
The true challenge of fighting those infections is time. In order to best treat their patients, doctors would like to know exactly which bacteria they are infected with, but the lost hours or days spent identifying the exact pathogen can make the road to recovery that much steeper. Sepsis, for instance, can develop so rapidly that mortality has been seen to increase by 9 percent per hour until treatment is given. Waiting two days may kill the patient, Park added.
For that reason, many hospital-acquired infections are treated presumptively, before they are definitively identified, using broad-spectrum antibiotics. These powerful combinations of potent drugs are often effective, but using them routinely raises the risk of deadly multidrug-resistant bacteria emerging.
The new QPIU technology promises to deliver better point-of-care diagnostics by reducing the time it takes to specifically identify bacteria, which may guide treatment, allowing doctors to prescribe the best drugs available to treat an infection and improving outcomes for people with hospital-acquired infections—though the effectiveness of the approach remains to be proven in future clinical trials.
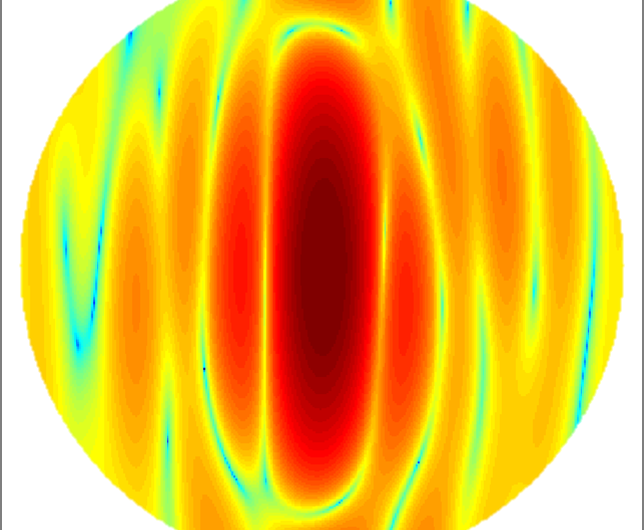
In their initial experiments, Park and his colleagues showed as a proof of principle that they could identify bacteria with high accuracy. They examined four different bacterial species (Listeria monocytogenes, Escherichia coli, Lactobacillus casei, and Bacillus subtilis). The first three are all pathogens known to infect humans through the food chain or via hospital-acquired infections. The fourth is a harmless bacteria used in laboratory research but of great interest because it is closely related to the deadly Bacillus anthracis, which is the base for Anthrax.
Under a microscope, all four of these rod-like bacteria look nearly identical. They would be virtually impossible to distinguish. The KAIST team, however, sorted them using the QPIU process with an accuracy greater than 94 percent.
The technique involves scattering light off the bacteria under the holographic microscope and then applying a mathematical function known as a Fourier Transform to the holographic images. The Fourier Transform allows them to define a distinct, fingerprint-like light scattering pattern for any given bacterial cell. They then applied software they designed to the analysis, which used a conventional approach to statistical classification known as machine learning—a sorting strategy based on pattern similarities that has been extensively used in applications like facial recognition software. This was the first time anyone had applied machine learning to Fourier Transform light scattering data, Park said.
They are now looking to extend their initial work to see if they can distinguish between several types of bacterial subgroups—to identify the most drug resistant or virulent strains from the innocuous ones.
In addition to helping in the clinic, the new method may be useful in the food industry or for homeland security applications. In principle, the approach could be scaled up to screen for contaminated food or suspicious packages.
More information: "Label-free identification of individual bacteria using Fourier transform light scattering," YoungJu Jo et al., Opt. Express 23, 15792-15805 (2015). www.osapublishing.org/oe/abstr … m?uri=oe-23-12-15792
Journal information: Optics Express , Optics Letters
Provided by Optical Society of America