April 29, 2015 report
Researchers deliver transcription factors into specific tissue of a living animal for the first time
A team of researchers affiliated with the University of California has for the first time, delivered transcription factors into specific tissue of a living animal. In their paper published in the journal Nature Materials, the team describes how they built on prior research to come up with a new technique that allowed for the breakthrough.
Transcription factors are proteins that are in charge of turning genes on and off—they bind to sections of DNA and regulate the process of transcription of DNA into mRNA and by extension, the proteins that are expressed. Scientists have long wished for a way to send transcription factors to specific parts of the body because it would allow for positive things to happen, such as boosting production of proteins that are known to help suppress tumors—all without permanently altering DNA. But finding a way to make it happen has been difficult because in order to work, transcription factors must make their way into cells, no easy feat because cells have built-in protection systems.
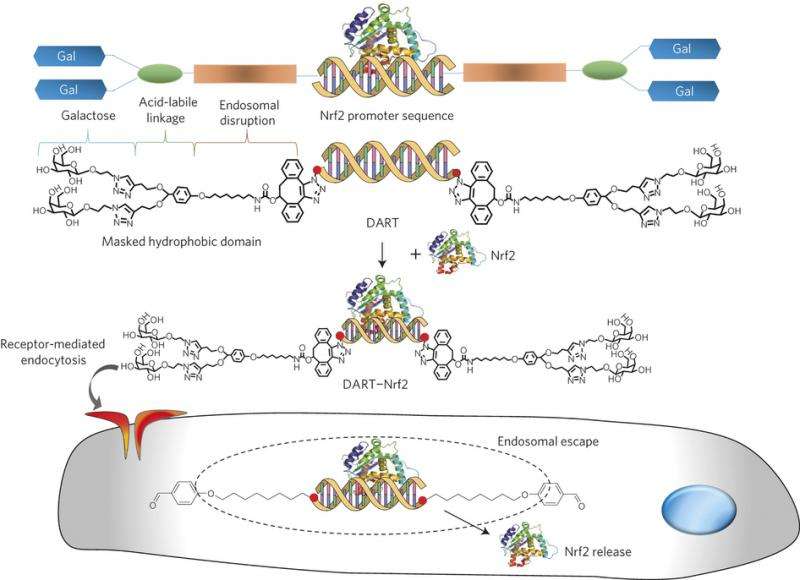
Four years ago a team of researchers made progress by gluing a transcription factor to a strand of DNA that matched those of targeted cells—they were able to successfully inject the transcription factor into living cells in a Petri dish. In this new effort, the researchers have expanded on that idea, they used a matched DNA strand as a type of scaffold to allow for carrying, in addition to the transcription factor, other molecules helpful in penetrating a cell's defenses, and also, some that help get the material to the part of the body being targeted: in this case, liver cells. The team calls the complex they made, DNA assembled recombinant transcription factor (DART).
In their initial tests, the researchers chose a transcription factor that has already been the subject of a lot of study, Nrf2. It is heavily involved in regulating genes that express proteins that help control inflammation and promote antioxidants. Mice were first injected with acetaminophen to cause damage to their livers—next, the DART was injected into some of the mice, but not others. Testing of the mice showed that the DART had indeed made its way to the liver, and better yet, the mice that had received it had livers that looked nearly identical to mice with healthy livers—those that did not receive the DART, of course showed severe liver damage.
More information: In vivo delivery of transcription factors with multifunctional oligonucleotides, Nature Materials (2015) DOI: 10.1038/nmat4269
Abstract
Therapeutics based on transcription factors have the potential to revolutionize medicine but have had limited clinical success as a consequence of delivery problems. The delivery of transcription factors is challenging because it requires the development of a delivery vehicle that can complex transcription factors, target cells and stimulate endosomal disruption, with minimal toxicity. Here, we present a multifunctional oligonucleotide, termed DARTs (DNA assembled recombinant transcription factors), which can deliver transcription factors with high efficiency in vivo. DARTs are composed of an oligonucleotide that contains a transcription-factor-binding sequence and hydrophobic membrane-disruptive chains that are masked by acid-cleavable galactose residues. DARTs have a unique molecular architecture, which allows them to bind transcription factors, trigger endocytosis in hepatocytes, and stimulate endosomal disruption. The DARTs have enhanced uptake in hepatocytes as a result of their galactose residues and can disrupt endosomes efficiently with minimal toxicity, because unmasking of their hydrophobic domains selectively occurs in the acidic environment of the endosome. We show that DARTs can deliver the transcription factor nuclear erythroid 2-related factor 2 (Nrf2) to the liver, catalyse the transcription of Nrf2 downstream genes, and rescue mice from acetaminophen-induced liver injury.
Journal information: Nature Materials
© 2015 Phys.org