New materials repel oil underwater, could better clean up oil spills

University of Wisconsin-Madison researchers have announced a significant step forward in the development of materials that can ward off oil—a discovery that could lead to new protective coatings and better approaches to cleaning up oil spills.
In a new paper in the journal Advanced Functional Materials, professor of chemical and biological engineering David Lynn and assistant scientist Uttam Manna describe new coatings that are extremely oil-repellant (or "superoleophobic") in underwater environments.
Lynn and Manna didn't initially set out to develop highly oil-repelling materials, but their work on fine-tuning the nano- and micro-scale structure of materials led to the unexpected finding.
Manna says a unique, layer-by-layer approach to fabricating thin, multi-layer polymer films allowed the researchers to manipulate both the chemistry and the topography of the material, yielding three-dimensional structures that are porous at the micrometer and nanometer scale.
Even when damaged or subjected to physical abrasion, these porous materials remain superoleophobic. They also held up in simulated seawater, lake water and in the presence of proteins and other surface-active contaminants, making them good candidates for a range of practical applications.
"(These features) address some of the problems with other types of 'superphobic' surfaces," says Lynn. "Once you put them out in the real world, they stop working."
In one test, the researchers coated a glass slide with the superoleophobic film and abraded a section of the coating with sandpaper. The damaged areas continued to repel droplets of oil, which remained suspended above the surface in an ellipsoid shape, just as effectively as the untouched areas. The researchers also placed the film in boiling water, scratched it, froze it, stuck tape to it and ripped the tape off. Even when they succeeded in damaging the material, the researchers found they didn't break down the porous structure that makes it oleophobic.
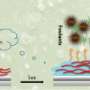
The materials can also be used to easily separate oil and water. In one test, researchers coated wire mesh with the material, then poured a mixture of water and motor oil through it. The water passed through and nearly all of the oil accumulated on top of the mesh, allowing it to be easily collected and recovered. The effectiveness of this simple process, and the ability to tune the properties of these materials, points to potential applications in all manner of situations where one chemical needs to be separated from another.
"We're not just talking about the potential to develop new tools to help clean up oil spills," Lynn says. "It could also be fragrances and pharmaceuticals, fine chemicals or other organic liquids."
The researchers say the next challenge is to translate the lessons learned from these new materials into a viable process for creating new oil-repellant coatings on larger scales.
"We'll find, over time, that there are better ways of making materials that have these same physical and chemical properties," Lynn says. "With the principles that we've uncovered, we can now go back and try to do on purpose what we discovered by accident—and find a way to make it scalable."
Journal information: Advanced Functional Materials
Provided by University of Wisconsin-Madison