February 5, 2015 report
New process allows for stronger, lighter, flexible steel

(Phys.org) —A trio of researchers with South Korea's Graduate Institute of Ferrous Technology has found a way to create a new low-density steel that is stronger, lighter and more flexible than the conventional steel that is used in so many manufacturing applications. In their paper published in the journal Nature, the team describes the process they used and their hopes that it might replace conventional steel in some applications sometime in the near future.
One of the biggest applications for steel is, of course in making cars, though its dominance has been slipping in recent years as engineers seek to find lighter substitutes. At issue is weight, steel is heavy because it is made mostly out of iron. Adding other lighter metals tends to make it less flexible or weaker. In this new effort, the researchers report that they have found a mix that allows for creating a low-density steel that is stronger and more flexible than much more expensive titanium alloys.
The secret, the team explains, lies in causing new structure shapes to be formed during the heating process and by using the right mix of ingredients. They used the traditional mix of iron, carbon, aluminum and manganese and then added some nickel. The nickel, they found reacted with the aluminum, creating nanometer sized B2 crystals that formed within and between the steel grains during the annealing process. To make sure the crystals were spread evenly among the metal, the team studied samples under an electron microscope. Chemists know that B2 crystals are resistant to shearing, thus steel with such crystals should be extremely strong, and that is what the researchers found when they tested their new alloy.

The researchers have already teamed with POSCO, one of the biggest steel makers in the world to see if the new kind of steel they have come up with might be usable in cars or even airplanes. The first step will be to see if the process is scalable, and if so, if it can be used to produce the new low-density steel at a cost that is competitive with conventional steel—the researchers are optimistic because all of the ingredients are low cost metals.
-

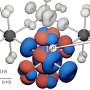
Annealed microstructure of high-specific-strength steel (HSSS). Fine FeAl-type B2 precipitates form during annealing in between the B2 stringer bands in steel matrix. Note that grain boundaries of recrystallized austenite crystals are visible in the image. The specimen was annealed for 15 min at 900 C. Credit: Hansoo Kim -

Annealed microstructure of high-specific-strength steel (HSSS). Fine FeAl-type B2 precipitates form during annealing in between the B2 stringer bands in steel matrix. Note that grain boundaries of recrystallized austenite crystals are visible in the image. The specimen was annealed for 15 min at 900 C. Credit: Hansoo Kim
More information: Brittle intermetallic compound makes ultrastrong low-density steel with large ductility, Nature 518, 77–79 (05 February 2015) DOI: 10.1038/nature14144
Abstract
Although steel has been the workhorse of the automotive industry since the 1920s, the share by weight of steel and iron in an average light vehicle is now gradually decreasing, from 68.1 per cent in 1995 to 60.1 per cent in 2011 (refs 1, 2). This has been driven by the low strength-to-weight ratio (specific strength) of iron and steel, and the desire to improve such mechanical properties with other materials. Recently, high-aluminium low-density steels have been actively studied as a means of increasing the specific strength of an alloy by reducing its density3, 4, 5. But with increasing aluminium content a problem is encountered: brittle intermetallic compounds can form in the resulting alloys, leading to poor ductility. Here we show that an FeAl-type brittle but hard intermetallic compound (B2) can be effectively used as a strengthening second phase in high-aluminium low-density steel, while alleviating its harmful effect on ductility by controlling its morphology and dispersion. The specific tensile strength and ductility of the developed steel improve on those of the lightest and strongest metallic materials known, titanium alloys. We found that alloying of nickel catalyses the precipitation of nanometre-sized B2 particles in the face-centred cubic matrix of high-aluminium low-density steel during heat treatment of cold-rolled sheet steel. Our results demonstrate how intermetallic compounds can be harnessed in the alloy design of lightweight steels for structural applications and others.
Journal information: Nature
© 2015 Phys.org