Arming nanoparticles for cancer diagnosis and treatment
UCD researchers have successfully manipulated nanoparticles to target two human breast cancer cell lines as a tool in cancer diagnosis and treatment.
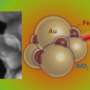
Coating nanoparticles with different substances allows their interaction with cells to be tuned in a particular way. For example, using an optically active particle like gold (Au) will provide excellent contrast in near infrared (NIR) imaging and, if heated, can actually destroy the surrounding tissue. This is called photothermal ablation therapy. Magnetically active particles like iron (Fe) can enable physical therapies by generating heat when exposed to alternating magnetic fields causing cell death (magnetic hyperthermia).
The UCD team led by Conway Fellows, Professor Gil Lee in the School of Chemistry and Chemical Biology and Professor Walter Kolch in Systems Biology Ireland, synthesised nanorods with a long iron segment coated with polyethylene glycol and a short gold tip coated with single layer of the protein, heregulin (HRG).
HRG is a growth factor that binds to and activates the ErbB family of protein receptors. ErbB2 is overexpressed in certain breast cancers and linked with poor prognosis. However, ErbB2 overexpression leads to enhanced sensitivity to certain drugs. The team believe that Fe-Au functionalised nanorods used in conjunction with these drugs could be useful in cancer treatment.
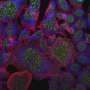
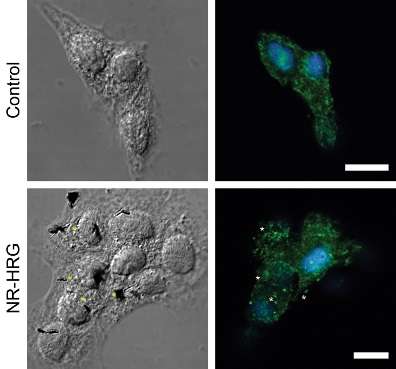
After characterising and tuning the interaction of the nanorods with the cells, the research team assessed how the cells respond to mechanical stimulation. To do this, they integrated an electromagnetic tweezers with an optical microscope and used a novel microfluidic chip to monitor the interaction of individual nanorods with two human breast cancer cell lines that express the ErbB family of receptors at different rates. When the HRG-nanorods bind to cancer cells expressing ErbB, they kick off a cascade of signalling events that lead to cell death.
"Using magnetic tweezers to stretch cells, we were able to further activate cell signalling pathways to trigger cell death. This was even more effective in causing cancer cell death than magnetic hyperthermia, the other therapeutic approach we assessed", explained Dr Devrim Kilinc, first author and research fellow in the Lee group.
"The results are a positive indication for nanoscale targeting and localised manipulation of cancer cells with a specific receptor profile."
More information: "Mechanochemical Stimulation of MCF7 Cells with Rod-Shaped Fe–Au Janus Particles Induces Cell Death Through Paradoxical Hyperactivation of ERK." Devrim Kilinc , Anna Lesniak , Suad A. Rashdan , Dhruv Gandhi , Agata Blasiak , Paul C. Fannin , Alex von Kriegsheim , Walter Kolch , and Gil U. Lee. Adv. Healthc. Mater. 2014, DOI: 10.1002/adhm.201400391
Provided by University College Dublin