The origins of multicellular life

The biological world around us is dominated by multicellular plants and animals. All of these intricate forms have evolved from far simpler, single celled ancestors.
What could explain the transition from single cells to cooperative groups, to groups of cells that put the prosperity of the whole group before the one? This is the essential question of how organisms evolved from single celled types and it is one of life's greatest mysteries.
In ground-breaking research reported in this week's edition of the science journal Nature, researchers from New Zealand, Germany and the USA report the real time evolution of life forms that have all the hallmarks of multicellular organisms.
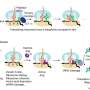
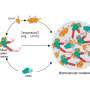
Beginning with single cells, the researchers show how simple cooperating groups of bacteria can reproduce via a life cycle that incorporates 'cheating' cells as a primitive germ line.
Cheats are cells that do not contribute to the integrity of the group, but still take advantage of the benefits of being part of a collective. An over abundance of cheating cells can cause the group to collapse.
Lead researcher Distinguished Professor Paul Rainey from the New Zealand Institute for Advanced Study (NZIAS) and Allan Wilson Centre at Massey University, and the Max Planck Institute for Evolutionary Biology in Germany, points out that the idea that cheats might be integrated into a life cycle is counter-intuitive.
"Cheats are typically viewed as the greatest impediment to the emergence of multicellular life because they collapse cooperating groups—the obvious thing to do is to get rid of them."
Joint first authors of the paper, Caroline Rose and Katrin Hammerschmidt, of the NZIAS, performed painstaking experiments over the course of five years in which they tested the idea that cheats might play a constructive role in evolution. They allowed simple microbial groups to evolve via a life cycle in which cheats were either embraced, or purged.
"When cheats were embraced we discovered something surprising," Dr Rose says. "Evolution saw a new kind of entity—a group comprised of two different cell states: cheating and cooperating cells. Evolution couldn't focus on just one state or the other; for lineages to persist, evolution had to see both types—it had to work on a developmental programme."
Dr Hammerschmidt explains: "When this happened, the groups became better adapted, but they did so at the expense of the individual cells that made up the groups. This might seem nonsensical, but it is precisely what is thought to happen during major evolutionary transitions: the higher (group) level subsumes the lower (cell) level, with the lower level eventually coming to work for the good of the collective. Nothing so remarkable happened when we performed the same experiment, but with a life cycle in which we got rid of cheats."
One of the most important outcomes of the work surrounds the origins of life cycles.
"Little is known", explains Professor Rainey, "but life cycles involving at least two different states are almost universal in the world of multicellular organisms. I suspect that this is because multiphase life cycles generate an organismal configuration that delivers to natural selection a machine-like entity with which it can really work.
"The emergence of these primordial life cycles holds the key to understanding some of biology's most profound problems: the origins of multicellularity; the origins of soma/germ differentiation, of reproduction, of development—even the origins of cancer."
More information: Hammerschmidt, K., Rose, C. J., Kerr, B. & Rainey, P. B. (2014). "Life cycles, fitness decoupling and the evolution of multicellularity." Nature 2014. DOI: 10.1038/nature13884
Journal information: Nature
Provided by Massey University