Billions of 'nanoreactors' inform materials design
Imagine building a chemical reactor small enough to study nanoparticles a billionth of a meter across. A billion times smaller than a raindrop is the volume of an E. coli cell. And another million times smaller would be a reactor small enough to study isolated nanoparticles. Add to that the challenge of making not just one of these tiny reactors, but billions of them, all identical in size and shape. Researchers at Cornell have done just that.
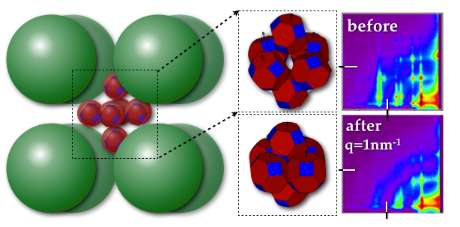
A team led by Tobias Hanrath, associate professor of chemical and biomolecular engineering, has demonstrated controlled fusion of semiconductor quantum dots within a nanoreactor cage of rusty particles.
The team arranged six lead selenide crystals within a framework of iron oxide (rust) spheres. They studied how the quantum dots within the nanoscale "rusty cage" interact, using X-rays at the Cornell High Energy Synchrotron Source (CHESS). These experiments allowed them to pinpoint specific interactions between particles in the box and thus pave the way for making novel materials with properties by design. The results, which could be applied to other materials, were published in Scientific Reports, Oct. 23.
They used CHESS to perform X-ray scattering on repeating units of these rusty boxes as they heated them up, watching what happens to the lead selenide in the center. With the scattering data acting like a high-definition movie, they could identify different stages of fusion of the lead selenide hexamers. This could lead to insight into getting specific functionalities out of these little-understood nanomaterials. Too much heat made the lead crystals sinter and fuse; not enough heat didn't pull them close enough together to interact.
Graduate student Ben Treml led the experiments; he synthesized the particles and assembled them into superlattices (lattices of nanocrystals, rather than atoms). The samples were studied at the D1 beam line of CHESS with co-author Detlef Smilgies, staff scientist, who helped Treml refine the experiments.
The results were verified with theoretical modeling by co-authors Paulette Clancy, professor of chemical and biomolecular engineering, and postdoctoral associate Binit Lukose.
More information: "Connecting the Particles in the Box - Controlled Fusion of Hexamer Nanocrystal Clusters within an AB6 Binary Nanocrystal Superlattice." Scientific Reports 4, Article number: 6731 DOI: 10.1038/srep06731
Journal information: Scientific Reports
Provided by Cornell University



















