Targeted nanoparticles that combine imaging with two different therapies could attack cancer, other conditions
Nanosystems that are 'theranostic'—they combine both therapeutic and diagnostic functions—present an exciting new opportunity for delivering drugs to specific cells and identifying sites of disease. Bin Liu of the A*STAR Institute of Materials Research and Engineering, and colleagues at the National University of Singapore, have created nanoparticles with two distinct anticancer functions and an imaging function, all stimulated on demand by a single light source. The nanoparticles also include the cell-targeting property essential for treating and imaging in the correct locations.
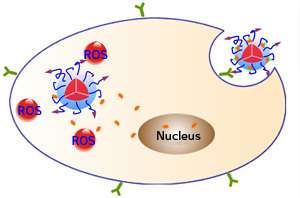
The system is built around a polyethylene-glycol-based polymer that carries a small peptide component that allows it to bind preferentially to specific cell types. The polymer itself serves as a photosensitizer that can be stimulated by light to release reactive oxygen species (ROS). It also carries the chemotherapy drug doxorubicin in a prodrug form.
The natural fluorescence of the polymer assists with diagnosis and monitoring of therapy as it shows where nanoparticles have accumulated. The ROS generated by light stimulation have a direct 'photodynamic' therapeutic activity, which destroys the targeted cells. The ROS additionally break the link between the polymer and the doxorubicin. Thus, cancer cells can be subjected to a two-pronged attack from the ROS therapy and the chemotherapy drug that is released within them (see image).
"This is the first nanoplatform that can offer on-demand and imaging-guided photodynamic therapy and chemotherapy with triggered drug release through one light switch," explains Liu, emphasizing the significance of the system.
The researchers demonstrated the power of their platform by applying it to a mixture of cultured cancer cells, some of which overexpressed a surface protein that could bind to the targeting peptide on the nanoparticles. Fluorescence imaging indicated that the nanoparticles were taken up by the target cells and that ROS and doxorubicin were released within these cells—all at significantly higher levels than in cells used as controls. The doxorubicin that was released in the cell cytoplasm readily entered the nucleus—its site of activity. Crucially, the combined therapy had a greater cytotoxic effect than any one therapy alone.
"The white light used in this work does not penetrate tissue sufficiently for in vivo applications," Liu explains, "but we are now attempting to use near-infrared laser light to improve the tissue penetration and move toward on-demand cancer therapy." She also suggests that with a few modifications, the system may be suitable for the diagnosis and treatment of other pathological processes including inflammation and HIV infection.
More information: Yuan, Y., Liu, J. & Liu, B. "Conjugated-polyelectrolyte-based polyprodrug: Targeted and image-guided photodynamic and chemotherapy with on-demand drug release upon irradiation with a single light source." Angewandte Chemie International Edition 53, 7163–7168 (2014). dx.doi.org/10.1002/anie.201402189
Journal information: Angewandte Chemie International Edition