September 16, 2014 feature
Nanocontainers for nanocargo: Delivering genes and proteins for cellular imaging, genetic medicine and cancer therapy
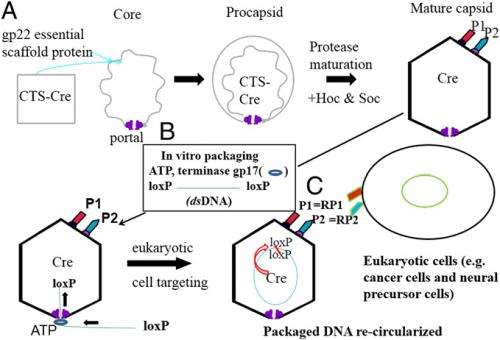
(Phys.org) —By loading any specific protein and nucleic acid into an icosahedral phage T4 capsid-based nanoparticle, the resulting cell delivery vehicle's ligands can bind to the surface of specific target tissues to deliver the protein/DNA cargo. (Icosahedral viral nanoparticles are evolutionary protein shells assembled in a hierarchical order that results in a stable protein layer and an inner space for accommodating nucleic acids and proteins; a capsid is the protein shell of a virus.) The technique has drug- and gene-delivery applications in human diseases, diagnostic and cellular imaging, and other medical areas. Recently, scientists at US Naval Research Laboratory, Washington, DC and University of Maryland at Baltimore packaged T4 nanoparticles in vivo with active cyclic recombination, or Cre, recombinase (a genetic recombination enzyme used to manipulate genome structure and control gene expression) and in vitro with fluorescent mCherry (a fluorescent protein used as a marker when tagged to molecules and cell components) expression plasmid DNA, and delivered these nanoparticles into cancer cells: When released into cells in the presence of both DNA and protein, the recombinase enhances mCherry expression by circularization (that is, changing the packaged linear DNA into a circular loop). The researchers state that this efficient and specific packaging into capsids and the unpackaging of both DNA and protein with release of the enzymatically altered protein/DNA complexes from the nanoparticles into cells have potential in numerous downstream applications such as genetic and cancer therapeutics.
Dr. Jinny L. Liu discussed the paper that she, Prof. Lindsay W. Black and their co-authors published in Proceedings of the National Academy of Sciences USA. "Icosahedral viral nanoparticles are essentially 100 nm by 80 nm nanocontainers that allow exogenous genetic material to be packaged in vitro through nucleic acid machinery that generally only allows linear DNA/RNA to be packaged through a portal channel," Liu tells Phys.org. "However, in vitro protein packaging is generally impossible, because for most viral nanoparticles there is no protein packaging machinery comparable to nucleic acid packaging machinery." While protein may be chemically cross-linked to the capsid inner surface, this is expected to lead to protein denaturation and loss of enzymatic activity.
That being said, nature has evolved solutions to this protein packaging conundrum. During in vivo viral capsid assembly, Liu explains, some bacterial viruses, or bacteriophages, target proteins within the procapsids before the nucleic acid is packaged so as to eject the proteins with the nucleic acid, thereby facilitating infection in conjunction with the nucleic acid. (A procapsid, or prohead, is an immature viral capsid structure formed in the early stages of self-assembly of some bacteriophages. Production and assembly of stable proheads is an essential precursor to bacteriophage genome packaging.) Only a few phages have well-characterized in vivo protein packaging systems, and phage T4 is the best characterized. "Prof. Black's lab at UMB and my lab at NRL have demonstrated that not only can a specific foreign enzyme – cyclic recombination (Cre) recombinase – be packaged into the capsid in vivo, but also that it is active within the capsid." This activity was demonstrated by showing the religation (the rejoining of two DNA strands or other molecules by a phosphate ester linkage) of packaged linear DNA flanked with two Cre recombination sites.
The paper shows that the substantial space within a T4 nanocontainer accommodates the active Cre enzyme along with exogenous DNA. "For potential applications, T4 can package up to 50 kb exogenous linear DNA containing full-length desired genes along with recombinases, either Cre or λ-red proteins, for specific homologous recombination within the chromosome," Liu notes. (Homologous recombination is a type of genetic recombination in which nucleotide sequences are exchanged between two similar or identical molecules of DNA.) "We expect that the cas9 enzyme could be encapsidated in a comparable way – and in fact, at least eight different proteins have been encapsidated in this manner. Through homologous recombination, our system can allow the corrected gene to replace the mutated gene in its original location within the chromosome or by precisely knocking out the overactive genes in stem cells." Liu points out that the T4 delivery vector is safer and better controlled than other viral delivery gene therapy, such as those delivering genes using infectious animal viral vectors to randomly insert the gene within the chromosome.
In their paper, the authors report that the T4 capsid NP gene expression and protein delivery system may be complementary to or used in conjunction with gene therapy based on RNA Cas and taran nuclease. (Cas genes code for proteins related to DNA loci containing short repetitions of base sequences known as Clustered Regularly Interspaced Short Palindromic Repeats, or CRISPRs.) "The T4 nanoparticle expression system can easily complement Cas9 and taran nuclease-based recombination by packaging the linear cas9, target-sgRNA plasmid DNA, and Cre recombinase – or even ligase, an enzyme that facilitates the joining of DNA strands – and deliver the resulting T4 nanoparticles into the recipient eukaryotic cells with high specificity employing SOC and HOC," Liu tells Phys.org. (SOC and HOC are dispensable T4 capsid proteins.) "By displaying the targeting ligands (binding molecules) onto the surface, the T4 capsid gene expression and protein system will be able to efficiently deliver the Cas9 and sgRNA plasmids together into the desired recipient cells. Relevant enzymatically-active proteins Cas9, lambda exonuclease, lambda beta protein and others can be delivered directly at the same time from the T4 nanoparticle."
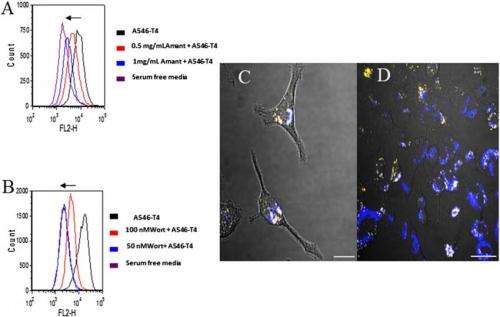
Liu adds that her lab has also been studying cell imaging and drug/gene delivery to eukaryotic cells using T4 tailless nanoparticles, which the researchers demonstrated can enter the eukaryotic cells without causing cell death.
A specific example of potential downstream drug and gene therapeutic applications resulting from the new approach is delivery of the toxic protein and linear plasmid that produces neutralizing peptides or antibodies into targeted cancer cells displaying specific cancer markers using high affinity SOC + HOC marker binding proteins on the surface of the capsids, while another example is to use the system for HIV gene therapy.
Liu adds that there are several pathways to use this system for gene therapy:
- Delivering T4 nanoparticles packaged with the recombinase (or ligase) and linear plasmid DNA to produce gp120 or interferon to generate or boost the immune response in patients
- Delivering T4 nanoparticles packaged with recombinase (ligase) and the linear soluble CD40 expression plasmid DNA into T lymphocytes or hematopoietic cells to block the infection of HIV-1
- Inhibiting RNA by delivering the engineered plasmid DNA that can produce decoy RNA for binding the viral sense DNA
- Inhibiting protein by delivering packaged anti-viral antibodies and anti-HIV antibody plasmid DNA
In addition to diagnostic and cellular imaging, the T4 nanoparticle gene-protein system can deliver repaired genes to correct human genetic diseases – for example, reversing adenosine deaminase (ADA) deficiency by introducing the protein-DNA complex to express ADA in stem cells. Other broad areas of research impacted by gene therapy technologies, such as genetic defects, cancer, neurological diseases in adults, and aging itself, may also benefit from this study.
Moving forward, the scientists want to develop more T4 procapsids packaging exonuclease and other recombinases along with engineered target DNA to demonstrate that the resulting T4 capsids can insert the gene into a stem cell line with a genetic deficiency. "In addition," Liu concludes, "we're working on adapting our system to deliver therapeutic peptides or antibodies to cells exposed to or infected by biothreat agents, such as protein toxins or viruses, efficient neutralization of toxin effects. The treatment and cure of cells and tissues exposed to such agents are of a great interest to our biodefense research community."
More information: Viral nanoparticle-encapsidated enzyme and restructured DNA for cell delivery and gene expression, Proceedings of the National Academy of Sciences USA, Published online before print August 26, 2014, doi:10.1073/pnas.1321940111
Journal information: Proceedings of the National Academy of Sciences
© 2014 Phys.org