Blocking cell division with two synergistic chemical inhibitors
(Phys.org) —The cycle of cell division—one cell splitting itself into two—is a crucial and complex process managed by finely tuned molecular machines. When working properly, cell division assures healthy growth. When running out of control, it can usher in cancer.
Blocking cell division in disease has been the target of researchers hoping to induce the death of abnormal cells before they become cancerous tumors. Finding the right chemical compound to inhibit cell division gone awry has proved difficult: Target the cell cycle too broadly and healthy cells will also suffer, as when chemotherapy hits all cells that divide rapidly, not just cancerous ones. Narrow the sights too tightly and the misbehaving machine churns on.
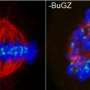
Now a team led by Randall King of Harvard Medical School has shown how two chemical inhibitors working together act better than either one alone, shutting down the dividing cell by stalling mitosis, one step in the cycle during which the cell copies and then lines up chromosomes properly so each daughter cell has a complete set.
The findings appear in Nature.
Working in animal and human cell lines, the scientists successfully disrupted two protein-protein interactions in one protein machine involved in mitosis, jamming the gears of cell division so long that the cells died. Certain types of cancer cells could be sensitive to these inhibitors because they already idle in mitosis longer than normal cells do before exiting this stage for the next step in the cell cycle.
"Simultaneous disruption of multiple interactions in a protein machine may be an interesting way to go in terms of trying to design future therapeutic strategies," said King, HMS professor of cell biology. "You're basically targeting one step in the pathway, but there's a lot of complexity in that one step. The idea is to disable the biochemical or enzymatic function by simultaneously targeting multiple sites."
King discovered the two inhibitors 10 years ago, in the very first screen conducted at the Institute of Chemistry and Cell Biology-Longwood Screening Facility at HMS. It was an unbiased chemical screen, set up with no assumptions about what they might find. Especially in the era before the discovery of RNA interference and its usefulness in silencing genes, scientists needed chemical tools that would perturb biological processes in other ways, so they could understand in detail how the mechanisms they were examining worked.
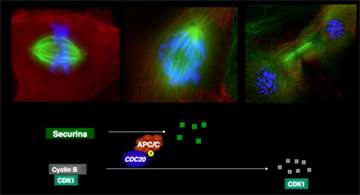
King's goal in 2004 was to fish through all the identified candidates from these early screens for chemical compounds that would somehow illuminate the cell cycle pathway and perhaps stymie one of its protein machines: the anaphase-promoting complex/cyclosome (APC/C). This protein complex marks certain proteins for degradation by the proteasome, the cell's waste-disposal site, before it can progress through mitosis.
If the APC/C doesn't tag these proteins with a protein called ubiquitin, the proteasome doesn't recognize them, they don't get discarded and mitosis cannot proceed, stalling the cell cycle before it can properly segregate its chromosomes for faithful division.
In 2010 King and his colleagues published a paper in Cancer Cell that described in detail how one of the inhibitors, called tosyl-L-arginine methyl ester (TAME), weakens the interaction between the APC/C and its critical activating protein, Cdc20. Degradation is blocked, but only partially. That means the cell cycle is delayed briefly, but still continues toward mitotic exit.
Now the scientists have shown how another compound, also discovered in the original 2004 chemical screen, binds in a pocket on Cdc20 that normally recruits the targets of APC/C. Called apcin (for APC inhibitor), it also delays mitosis, but only by a little bit.
Together, TAME and apcin slow mitosis to a crawl. The cell dies before it can leave mitosis.
The chemical compounds are not themselves drugs, but King says the findings provide a conceptual framework that may also be useful for inhibiting other protein machines. Until relatively recently, going after protein-protein interactions was considered a bad bet. Some protein-protein interactions are weak, with binding partners that are easily disturbed, making them harder to grab, King said.
His team's current work could conceivably add to success stories in protein-protein interactions.
"From a drug discovery standpoint, we think that perturbing the APC/C may be useful in certain therapeutic contexts related to cancer primarily and to the nervous system as well," King said. "One of the things we're examining now is how these compounds affect the ability of cancer cells to divide, especially because cancer cells have defects in some of the pathways that control chromosome segregation."
King said his lab is pursuing more potent forms of apcin based on the structure of the APC/C complex.
"Having the chemical tools now, we can easily screen cancer cells and ask if there are different lines that are particularly sensitive to this type of intervention," he said.
More information: "Synergistic blockade of mitotic exit by two chemical inhibitors of the APC/C." Katharine L. Sackton, et al. Nature (2014) DOI: 10.1038/nature13660. Received 07 August 2013 Accepted 04 July 2014 Published online 24 August 2014
"Pharmacologic Inhibition of the Anaphase-Promoting Complex Induces A Spindle Checkpoint-Dependent Mitotic Arrest in the Absence of Spindle Damage." Xing Zeng, Frederic Sigoillot, Shantanu Gaur, Sungwoon Choi, Kathleen L. Pfaff, Dong-Chan Oh, Nathaniel Hathaway, Nevena Dimova, Gregory D. Cuny, Randall W. King. DOI: dx.doi.org/10.1016/j.ccr.2010.08.010
Journal information: Cancer Cell , Nature
Provided by Harvard Medical School