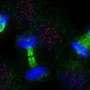
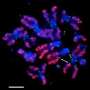
Faithful cell division requires tightly controlled protein placement at the centromeres
From fertilized egg to adult, the cells of the human body go through an astronomical number of divisions. During division of any of the body's roughly 30 trillion cells, DNA from the initial cell must be split precisely between the two resulting cells. Critical to successful cell division is the integrity of the centromere—a region of DNA on each chromosome where the cell division machinery attaches to segregate the chromosomes. For the segregation machinery to recognize this region, it must contain many copies of a pivotal protein known as CENP-A.
Each time the cell makes a copy of its DNA in preparation for cell division, it needs to ensure that the centromeres of the new and old DNA strand get re-filled with CENP-A. Although scientists have known that the centromere replenishes its stock of CENP-A deposits during a phase of the cell cycle referred to as G1, exactly how this process is regulated had remained a mystery.
Now work by Whitehead Institute Member Iain Cheeseman and Kara McKinley, a graduate student in Cheeseman's lab has identified the molecular controls that ensure that CENP-A deposition at centromeres occurs in the right place at precisely the right time. Their findings are described in the current issue of the journal Cell.
In preparation for mitosis, the cell's DNA is copied and condensed into paired chromosomes. Thin protein fibers, called microtubules stretch out from points on opposite sides of the cell and latch onto a protein complex, called the kinetochore, which is anchored by CENP-A molecules at the centromere. As mitosis progresses, the microtubules align the chromosomes along the mid-line of the cell, then shorten and pull the chromosome pairs at their centromeres to opposite sides of the cell.
The fate of the cell depends on this process going smoothly. If the chromosomes have too many or too few centromeres, or the centromeres are located in the wrong place, proper chromosome segregation fails, and the cell either dies or becomes diseased. Because the centromere's integrity is so vital, its precise location is amazingly maintained each time cells divide, and passed on through multiple generations.
In her research, McKinley found that two kinases—Plk1 and CDK—work in separate steps to ensure that CENP-A replenishment proceeds correctly. For CENP-A to fill in all of the gaps in the centromere, both steps must work correctly. By defining the ways in which these kinases work, McKinley was able to disrupt the precise timing of CENP-A replenishment and cause severe problems for chromosome segregation.
"The assumption was that the timing of CENP-A replenishment had to be important for centromere function because it was so tightly controlled. Now we have finally been able to test that assumption," says McKinley.
"Kara's work has defined the basis for how the CENP-A deposition process is controlled," says Cheeseman, who is also associate professor of biology at Massachusetts Institute of Technology. "Since this step is such a central part of being a centromere, her work is a fundamental advance in how we think about this as a process."
More information: "Polo-like kinase 1 licenses CENP-A deposition at centromeres" Cell, July 17, 2014.
Journal information: Cell
Provided by Whitehead Institute for Biomedical Research