New technique for identifying gene-enhancers
An international team led by researchers with the Lawrence Berkeley National Laboratory (Berkeley Lab) has developed a new technique for identifying gene enhancers - sequences of DNA that act to amplify the expression of a specific gene – in the genomes of humans and other mammals. Called SIF-seq, for site-specific integration fluorescence-activated cell sorting followed by sequencing, this new technique complements existing genomic tools, such as ChIP-seq (chromatin immunoprecipitation followed by sequencing), and offers some additional benefits.
"While ChIP-seq is very powerful in that it can query an entire genome for characteristics associated with enhancer activity in a single experiment, it can fail to identify some enhancers and identify some sites as being enhancers when they really aren't," says Diane Dickel, a geneticist with Berkeley Lab's Genomics Division and member of the SIF-seq development team. "SIF-seq is currently capable of testing only hundreds to a few thousand sites for enhancer activity in a single experiment, but can determine enhancer activity more accurately than ChIP-seq and is therefore a very good validation assay for assessing ChIP-seq results."
Dickel is the lead author of a paper in Nature Methods describing this new technique. The paper is titled "Function-based identification of mammalian enhancers using site-specific integration." The corresponding authors are Axel Visel and Len Pennacchio, also geneticists with Berkeley Lab's Genomics Division. (See below for a complete list of authors.)
With the increasing awareness of the important role that gene enhancers play in normal cell development as well as in disease, there is strong scientific interest in identifying and characterizing these enhancers. This is a challenging task because an enhancer does not have to be located directly adjacent to the gene whose expression it regulates, but can instead be located hundreds of thousands of DNA base pairs away. The challenge is made even more difficult because the activity of many enhancers is restricted to specific tissues or cell types.

"For example, brain enhancers will not typically work in heart cells, which means that you must test your enhancer sequence in the correct cell type," Dickel says.
Currently, enhancers can be identified through chroma¬tin-based assays, such as ChIP-seq, which predict enhancer elements indirectly based on the enhancer's association with specific epigenomic marks, such as transcription factors or molecular tags on DNA-associated histone proteins. Visel, Pennacchio, Dickel and their colleagues developed SIF-seq in response to the need for a higher-throughput functional enhancer assay that can be used in a wide variety of cell types and devel-opmental contexts.
"We've shown that SIF-seq can be used to identify enhancers active in cardiomyocytes, neural progenitor cells, and embryonic stem cells, and we think that it has the potential to be expanded for use in a much wider variety of cell types," Dickel says. "This means that many more types of enhancers could potentially be tested in vitro in cell culture."
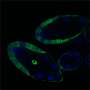
In SIF-seq, hundreds or thousands of DNA fragments to be tested for enhancer activity are coupled to a reporter gene and targeted into a single, reproducible site in embryonic cell genomes. Every embryonic cell will have exactly one potential enhancer-reporter. Fluorescence-activated sorting is then used to identify and retrieve from this mix only those cells that display strong reporter gene expression, which represent the cells with the most active enhancers.
"Unlike previous enhancer assays for mammals, SIF-seq includes the integration of putative enhancers into a single genomic locus," says co-corresponding author Visel. "Therefore, the activity of enhancers is assessed in a reproducible chromosomal context rather than from a transiently expressed plasmid. Furthermore, by making use of embryonic stem cells and in vitro differentia¬tion, SIF-seq can be used to assess enhancer activity in a wide variety of disease-relevant cell types."
Adds co-corresponding author Pennacchio, "The range of biologically or disease-relevant enhancers that SIF-seq can be used to identify is limited only by currently available stem cell differentiation methods. Although we did not explicitly test the activity of species-specific enhancers, such as those derived from certain classes of repetitive elements, our results strongly suggest that SIF-seq can be used to identify enhancers from other mammalian genomes where desired cell types are difficult or impossible to obtain."
The ability of SIF-seq to use reporter assays in mouse embryonic stem cells to identify human embryonic stem cell enhancers that are not present in the mouse genome opens the door to intriguing research possibilities as Dickel explains.
"Human and chimpanzee genes differ very little, so one hypothesis in evolutionary genomics holds that humans and chimpanzees are so phenotypically different because of differences in the way they regulate gene expression. It is very difficult to carry out enhancer identification through ChIP-seq that would be useful in studying this hypothesis," she says. "However, because SIF-seq only requires DNA sequence from a mammal and can be used in a variety of cell types, it should be possible to compare the neuronal enhancers present in a large genomic region from human to the neuronal enhancers present in the orthologous chimpanzee region. This could potentially tell us interesting things about the genetic differences that differentiate human brain development from that of other primates."
More information: test.nature.com/nmeth/journal/ … t/pdf/nmeth.2886.pdf
Journal information: Nature Methods
Provided by Lawrence Berkeley National Laboratory