March 28, 2014 feature
Physicists propose explanation for metals behaving badly
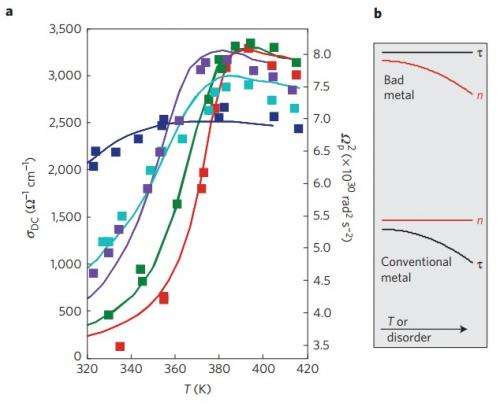
(Phys.org) —One of the defining properties of metals is that, the hotter the metal, the worse it conducts electricity. But while most metals obey this inverse relationship between temperature and conductivity in a straightforward way as predicted by theory, other metals do not. At high temperatures, the electrons in these so-called "bad metals" ought to violate Heisenberg's uncertainty principle so that the metals no longer conduct electricity. However, experiments have shown that these metals do continue to conduct electricity at high temperatures. The failure of current models to explain this behavior is a central problem in condensed matter physics, a field that deals with understanding the physical properties of materials.
Now in a new paper published in Nature Physics, Shriram Ramanathan, Professor at Harvard University, along with Rafael Jaramillo (now at MIT) and Sieu D. Ha at Harvard, as well as D. M. Silevitch at the University of Chicago, have proposed an explanation of the origins of bad metal conductivity that obeys the uncertainty principle. According to the explanation, the number of electrons that are free to conduct electricity in bad metals is constantly changing—an effect that was once considered to involve disappearing electrons.
Disappearing electrons
"Metals conduct electricity because they contain electrons that are free to move through the material," Jaramillo told Phys.org. "For more than a century, it was thought that the number of free electrons in a metal remains fixed for a given material. However, in the 1980s so-called 'bad metals' were discovered that all had similar chemical and atomic structure. By the Heisenberg uncertainty principle, stimuli such as temperature or induced disorder were expected to destroy electrons in such materials, but this was not found. Instead, in bad metals the electrons seemed to reversibly disappear and reappear as stimuli were applied and removed.
"It was later hypothesized that this phenomenon occurs in bad metals by temporarily shifting electrons to hidden states in response to stimuli. In the hidden states, the electrons are effectively stowed away and are no longer free to move as they would in normal metals. When the stimuli are removed, electrons are returned from the hidden states. This process is akin to extreme weather conditions that cause people to stay inside their homes, reducing the sidewalk foot traffic outside and keeping people safe from the elements. In this analogy, the elements cause people to scatter off of objects (and each other), and fewer people on the sidewalk makes walking easier for those that remain."
Scattering to the limit
Similar to how strong winds blow pedestrians around, high temperatures cause the electrons in a metal to scatter more rapidly than they do at lower temperatures. The increased disorder causes a decrease in the metal's conductivity. This effect is called the scattering rate hypothesis and is part of the Drude model of electrical conduction, which has been used to describe metal conductivity for more than a century.
According to Heisenberg's uncertainty principle, this inverse relationship between conductivity and temperature cannot continue indefinitely. When the temperature exceeds a certain point, the electrons are predicted to scatter off one another so quickly that they should no longer have a well-defined energy. So the uncertainty principle places a limit on the time between scattering events (called either the scattering lifetime or the mean free time between collisions), and thus on a metal's maximum temperature.
For most metals, this limit isn't a problem because they start to melt long before they reach this high temperature. Other metals, called saturating metals, don't melt but still obey the limit because their conductivity saturates as they approach the limit, validating that the conductivity-temperature relationship does not continue indefinitely.
However, experiments have shown that bad metals can exceed this temperature without melting or exhibiting conductivity saturation. Instead, the bad metals remain conductors even at high temperatures at which the scattering hypothesis predicts violation of the uncertainty principle.
Fewer free electrons
If, as Jaramillo explained above, bad metal behavior is caused by electrons that seem to disappear and reappear, what causes these disappearances?
In the new paper, the physicists proposed an answer to this question by performing experiments on a new class of bad metals, the rare-earth nickelates—in particular, SmNiO3 (samarium nickelate). In their experiments, the scientists grew a series of samples of samarium nickelate by high-pressure sputtering, controlling the amount of disorder in each sample by varying the sputtering pressure.
Then the researchers measured the conductivity of each sample at different temperatures, and also compared the results to the conductivities of conventional metals from published data.
The physicists found that temperature (and disorder) does not affect nickelates in the same way as it affects conventional metals. Whereas in conventional metals, changes in temperature and disorder affect the scattering rate of electrons as explained above, in nickelates, changes in temperature and disorder produce continuous shifts in the number of electrons that are free to conduct electricity (the spectral weight).
This finding leads to the most important result of the paper: Redistributions in the nickelates' spectral weights enable them to remain conductors at high temperatures while preserving the Drude model and without violating the uncertainty principle. Going back to the analogy of pedestrians bumping into each other on the sidewalk, the fewer pedestrians (electrons), the less frequently they bump into each other. The time between electron scattering events is long enough to not violate the uncertainty principle.
The researchers' observations suggest that the changes in spectral weight occur due to interactions between electron-phonon interactions in nickelates. However, they caution that it will be challenging to develop a theory that fully describes these interactions.
Bad metals, promising futures
Nickelates aren't the only materials known to exhibit spectral weight shifting; the effect is also seen in Mott-Hubbard materials, which also behave slightly differently than conventional theories predict. By showing that bad metal conductivity can be explained by the shifting of spectral weight in nickelates, the results here provide a deeper understanding of electrical conductivity and help explain why some metals don't behave as obediently in accordance with standard theories as other metals do.
"Bad metal behavior seems to go hand-in-hand with some of the most poorly understood but technologically promising phenomena in physics, such as high-temperature superconductivity and colossal magnetoresistance," Jaramillo said. "Many of the same materials that exhibit these effects also turn out to be bad metals. Any advance that sheds light on bad metals therefore impacts a large swath of solid state physics, including a number of technologically promising materials.
"On a more fundamental level, understanding metals formed the core of 20th century solid state physics. From Drude's theory, to quantum mechanics and Bloch states, and culminating in Lev Landau's profound Fermi liquid theory, physicists have probably had a deeper understanding of metals than any other solid state. Therefore, an outstanding puzzle like bad metals is galling and really calls out for deeper understanding."
In the future, the physicists plan to study bad metals in more detail to better understand their unusual behavior.
"Future research directions would include investigating the role of strain and chemical disorder in bad metals," Jaramillo said. "For example, it would be fascinating to compare the response of a bad metal to different types of disorder, such as structural disorder and ion implantation."
More information: R. Jaramillo, et al. "Origins of bad-metal conductivity and the insulator-metal transition in the rare-earth nickelates." Nature Physics. DOI: 10.1038/NPHYS2907
Journal information: Nature Physics
© 2014 Phys.org. All rights reserved.




















