Researchers identify key enzyme found in bacteria responsible for heart valve disease
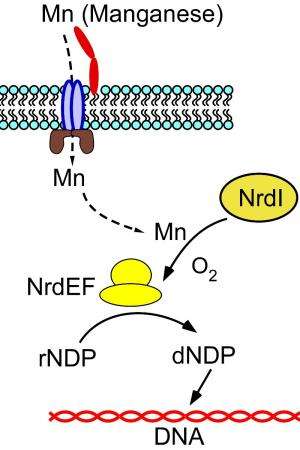
A disease-causing bacterium found in the mouth needs manganese, a trace mineral, in order to cause a serious heart infection, according to a preclinical study led by researchers at Virginia Commonwealth University Philips Institute for Oral Health Research in the School of Dentistry.
The findings, which may solve a longstanding mystery of why some bacteria need manganese to cause disease, provide possible new targets for antibiotics.
Researchers from VCU and MIT have been studying the bacterium Streptococcus sanguinis, which lives in the mouth, to understand its role in infective endocarditis, a heart valve disease. The infection is hard to treat and can be deadly – killing more than 20 percent of the people who contract it.
Researchers have known for some time that several types of bacteria responsible for serious infections – including S. sanguinis – need more manganese than others to grow normally.
In joint studies published this week in the Journal of Biological Chemistry, researchers showed that an enzyme that provides the building blocks needed for making DNA requires manganese to do its job. When the VCU team eliminated that enzyme or a second protein that attaches the manganese to the enzyme, then the bacterium could no longer cause endocarditis, nor survive within the animal model. The MIT team carefully examined the activity of the purified enzymes and determined the function of each. The VCU-MIT study is the first of its kind to test the importance of these enzymes for causing any disease.
Understanding the importance of manganese in the cell has been key to learning the best way to target the bacterium and stop it from causing disease, according to corresponding author Todd Kitten, Ph.D., associate professor at the Phillips Institute for Oral Health Research at the VCU School of Dentistry.
"The best antibiotics attack parts of a bacterium that are critical for bacterial survival, but are not found in human cells," Kitten said.
"The manganese-requiring enzyme meets both requirements because these bacteria need it to survive and humans use a very different, iron-containing enzyme to make DNA building blocks. It is the manganese requirement that makes the bacterial proteins good targets," he said.
Kitten added that humans have very little manganese in their bodies, so these bacteria require specialized systems to take in enough manganese to survive. These uptake systems are not found in humans. The team is in the early stages of a collaboration with Glen Kellogg, Ph.D., associate professor in the Department of Medicinal Chemistry at the VCU School of Pharmacy, to create designer drugs to attack the manganese uptake system in these bacteria.
Down the road, it could be possible to target several other disease-causing bacteria that also have this enzyme and likely need it to cause disease, including MRSA; the flesh-eating bacterium, Streptococcus pyogenes; and the bacterium that causes anthrax.
The team is also examining whether manganese has other activities in these bacteria that might be equally important.
The research builds on the previously published reports of other VCU researchers. A 1995 study published in Infection and Immunity led by Francis Macrina, Ph.D., currently vice president for research at VCU, showed for the first time that a protein that turned out to be a manganese uptake protein was necessary for causing disease.
In the years since, dozens of researchers have discovered similar proteins in other disease-causing bacteria. This study also builds on work done by a collaborative VCU group that was the first to determine the DNA sequence of S. sanguinis.
Journal information: Journal of Biological Chemistry , Infection and Immunity
Provided by Virginia Commonwealth University


















