Embryonic stem cells: Reprogramming in early embryos
An Oregon Health & Science University scientist has been able to make embryonic stem cells from adult mouse body cells using the cytoplasm of two-cell embryos that were in the "interphase" stage of the cell cycle. Scientists had previously thought the interphase stage—a later stage of the cell cycle—was incapable of converting transplanted adult cell nuclei into embryonic stem cells.
The findings by OHSU's Shoukhrat Mitalipov, Ph.D., and his team could have major implications for the science of generating patient-matched human embryonic stem cells for regenerative medicine. Human embryonic stem cells are capable of transforming into any cell type in the body. Scientists believe stem cell therapies hold promise for someday curing or treating a wide range of diseases and conditions—from Parkinson's disease to cardiac disease to spinal cord injuries—by replacing cells damaged through injury or illness.
Mitalipov's findings will be published March 26 in the online edition of Nature. If the new findings in mice hold true for humans, it could significantly help efforts to make rejection-proof human embryonic stem cells for regenerative therapies. That's because embryonic cells that Mitalipov's team used for reprogramming—cells in the "interphase" stage—are more accessible than the traditional egg cells that are in short supply. Scientists previously had believed embryonic stem cells were capable of being produced only using the metaphase stage of egg cytoplasm.
Embryonic stem cells can be made using a process called somatic cell nuclear transfer, or SCNT, in which the nucleus from an adult cell is transferred into the cytoplasm of an unfertilized egg cell. The cytoplasmic machinery then "reprograms" that nucleus and cell into becoming an embryonic stem cell capable of transforming to any type of cell in the body.
"It has always been thought that this capacity for reprogramming ended with metaphase," said Mitalipov, senior scientist at OHSU's Oregon National Primate Research Center. "Our study shows that this reprogramming capacity remains in the later embryonic cell cytoplasm even during interphase. It looks like the factors continue working and they efficiently reprogram the cells—just as they do in metaphase."
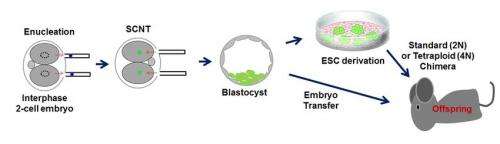
Many scientists have attempted to reprogram cells by interphase cytoplasm. Mitalipov and his team found success by carefully synchronizing the cell cycles of the adult cell nucleus and the recipient embryonic cytoplasm. Both had to be at an almost identical point in their respective cell cycles for the process to work, Mitalipov said.
"That was the secret," Mitalipov said. "When we did that matching, then everything worked."
Mitalipov said the next step to further his research will be to test the process in rhesus macaques.
Mitalipov has become a world scientific leader in embryonic stem cell research and in somatic cell nuclear transfer. He recently was named the director of a newly created research center at OHSU—the Center for Embryonic Cell and Gene Therapy. The center will help Mitalipov and his team accelerate their research, with expanded support from private philanthropy.
More information: Paper: dx.doi.org/10.1038/nature13134
Journal information: Nature
Provided by Oregon Health & Science University


















