Packaging insulin into a pill-friendly form for diabetes treatment
Since insulin's crucial discovery nearly a century ago, countless diabetes patients have had to inject themselves with the life-saving medicine. Now scientists are reporting a new development toward a long-sought insulin pill that could save millions the pain of daily shots. Published in the ACS journal Biomacromolecules, the advance could someday not only eliminate the "ouch" factor, but also get needle-wary—and weary—patients to take their medicine when they should.
Sanyog Jain and colleagues explain that patients with diabetes sometimes skip doses or stop taking their insulin because the injections can be painful. But doing so puts their health in danger. An estimated 347 million people globally (about 26 million in the U.S.) are living with diabetes. In the U.S., more than a quarter of these patients are taking some kind of insulin therapy. For years, researchers have sought a way to transform delivery of this therapy from a shot to a pill, but it has been a challenge. The body's digestive enzymes that are so good at breaking down food also break down insulin before it can get to work. In addition, insulin doesn't get easily absorbed through the gut into the bloodstream. To overcome these hurdles, Jain's team combined two approaches to shield insulin from the digestive enzymes and then get it into the blood.
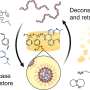
They packaged insulin in tiny sacs made of lipids, or fats, called liposomes, which are already used in some treatments. Then, they wrapped the liposomes in layers of protective molecules called polyelectrolytes. To help these "layersomes" get absorbed into the bloodstream, they attached folic acid, a kind of vitamin B that has been shown to help transport liposomes across the intestinal wall into the blood. In rats, the delivery system lowered blood glucose levels almost as much as injected insulin, though the effects of the layersomes lasted longer than that of injected insulin.
More information: "Improved Stability and Antidiabetic Potential of Insulin Containing Folic Acid Functionalized Polymer Stabilized Multilayered Liposomes Following Oral Administration" Biomacromolecules, Article ASAP. DOI: 10.1021/bm401580k
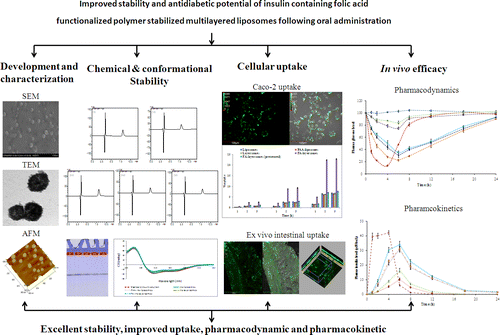
Abstract
The present study reports the folic acid (FA) functionalized insulin loaded stable liposomes with improved bioavailability following oral administration. Liposomes were stabilized by alternating coating of negatively charged poly(acrylic acid) (PAA) and positively charged poly(allyl amine) hydrochloride (PAH) over liposomes. Furthermore, folic acid was appended as targeting ligand by synthesizing folic acid–poly(allyl amine) hydrochloride conjugate. The insulin entrapped within the freeze-dried formulation was found stable both chemically as well as conformationally and developed formulation exhibited excellent stability in simulated biological fluids. Caco-2 cell and ex vivo intestinal uptake studies revealed higher uptake of folic acid functionalized layersomes in comparison with their plain counterparts. In vivo pharmacodynamic and pharmacokinetic studies further revealed almost double hypoglycemia and approximately 20% relative bioavailability in comparison with subcutaneously administered standard insulin solution. Overall the proposed strategy is expected to contribute significantly in the field of designing ligand-anchored, polyelectrolyte-based stable systems in drug delivery.
Journal information: Biomacromolecules
Provided by American Chemical Society