How glow-in-the-dark jellyfish revolutionised plant biology

What happens inside plant cells? How can we see proteins in living cells that aren't even visible with a microscope? This was a problem in plant cell biology until the discovery of a fluorescent jellyfish and the isolation of the protein responsible for its green glow. Over the past half a century since the discovery of green fluorescent protein (GFP) biologists have gained the tools necessary to illuminate the microscopic world inside of a living cell.
Three scientists involved in the discovery and development of GFP received the Nobel Prize in Chemistry in 2008, reflecting how this work has revolutionised cell biology research. The protein and its derivatives have been harnessed as essential tools for studying life at a protein level. No longer confined to the jellyfish species Aequorea victoria, GFP can be found inside cells in research labs across the globe.
Natural glow
In the early 1960s Osamu Shimomura arrived from Japan at Princeton University to study naturally glowing (bioluminescent) jellyfish. The species Aequorea victoria emitted a bright green glow and became the focus of their research. Several scientists including Shimomura travelled to Washington State where they used nets to collect, by hand, approximately 10,000 jellyfish, separating the outer ring containing the green light emitting segments and discarding the rest. They then ground the harvested parts and passed them through a cheese cloth to form what the team referred to as jellyfish "squeezate". From this they isolated two proteins. The first was aequorin, which required calcium to produce bioluminescence. Unlike aequorin, the second protein required activation with ultraviolet (UV) light for the fluorescence. This protein was green fluorescent protein (GFP). The capacity of GFP to function without requiring other interacting molecules was a novel observation and over the following decades its potential for use in research was realised.
The gene for GFP was cloned from the jellyfish DNA in 1992 by Douglas Prasher. Two years later Martin Chalfie inserted this gene into the cells of a bacterium, E. coli and the roundworm C. elegans. When the cells were exposed to UV light they emitted a green fluorescence, as the protein had done on its own in a test tube in Shimomura's laboratory.
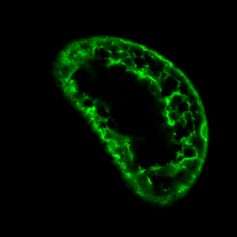
This initial work proved the concept that the gene for GFP could be inserted into the genetic code of different species, who can then be made to make their own GFP. Mutated variants of the GFP molecule were developed by Roger Tsien to produce different wavelengths of light and therefore different colours of fluorescence. Processes that were never seen before, quite literally, illuminated and able to be analysed as they happen inside living cells.
Research methods employing GFP have become one of the most widely used by plant cell biologist. Combined with the other techniques such as fluorescent stains and a powerful range of microscopes, GFP has enabled us to build a clearer picture of protein function in cells than ever before. As with the first experiments using bacteria and flatworms, plant biologists use the GFP gene fused to genes for proteins of interest and insert this DNA into the plant cells. As this is carried out in living tissue, the movement of the protein inside of the cell, as well as the initial location, can be imaged and analysed. For example, we can label membrane proteins in order to see and understand how cellular membranes move in real time.

With these techniques research is building an increasingly complete picture of the intricate, immensely complex and coordinated protein activity constantly occurring inside of cells.
Source: The Conversation
This story is published courtesy of The Conversation (under Creative Commons-Attribution/No derivatives).
![]()