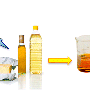New process targets valuable green chemicals

(Phys.org) —Milk cartons, plastic bags, car seats... the list of items made from petroleum extends far beyond fuel, increasing our reliance on limited fossil resources. That's why researchers in the Great Lakes Bioenergy Research Center (GLBRC) are developing "green" compounds that can be used to manufacture everyday products in addition to renewable replacements for oil and gasoline.
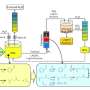
In the Department of Chemical and Biological Engineering, Steenbock Professor and Michel Boudart Professor James Dumesic leads research on developing such green chemicals from cellulosic—or non-food—plant materials like grasses, wood and crop residues. Dumesic and his team recently published a paper in Chemical Communications demonstrating a new, highly efficient process for converting biomass-derived feedstocks into linear alpha olefins (LAOs). The process is so commercially promising that the researchers have already filed a patent application with the Wisconsin Alumni Research Foundation (WARF).
While the term LAO may sound unfamiliar, it refers to the building blocks that form the chemical basis for a host of items that most of us use every day, like plastics, beauty products and detergents. "Our process produces the most valuable type of olefins from biomass, both in terms of product quality and market price," says Dong Wang, a graduate research assistant in the Dumesic lab and the paper's first author. "What's great about LAOs compared to other types of olefins is that they lend increased flexibility, strength or biodegradability to the products they are used to make."
U.S. demand for LAOs in 2007 was 3.4 million metric tons, and that figure is still on the rise. Existing LAO production technologies use petrochemicals as a starting material. The new process has demonstrated extraordinary advantages for both efficiency and sustainability. In addition to manufacturing bio-based chemicals, the Dumesic lab has already found its catalytic process could also be used to produce a green replacement for diesel fuel. "This paper is a step forward in laying the scientific foundation for producing renewable LAOs from biomass," says George Huber, a professor of chemical and biological engineering. "One of the key challenges of the 21st century is figuring our how we can end our dependence on petroleum oil. The chemical industry is completely dependent on petroleum as a feedstock to make chemicals, and companies are looking for alternatives due to rising oil prices and a desire to make the chemical industry greener. The Dumesic group's process will allow us to convert biomass into higher value commodity chemicals."

A unique feature of the new process is that it causes reaction compounds to separate out into layers, like an un-shaken salad dressing bottle, making it easy for researchers to extract the final product. In addition, the main catalyst used in the process is both inexpensive and non-toxic—a dual advantage when it comes to industrial chemical production.
Sikander Hakim, a postdoctoral research associate with the National Science Foundation Engineering Research Center for Biorenewable Chemicals (CBiRC) and an author on the paper, says the new process could also enhance industrial efficiency by giving manufacturers the control they need to produce only the specific molecules they are interested in. "With traditional technologies, you cannot simply increase production of a single type of olefin that happens to be in high demand," says Hakim. "You must instead produce the entire spectrum of molecules, but then you are stuck with the less valuable fractions that must be disposed of—by selling them at a discounted price, for example."
While significant time and energy are usually required to separate out the desired molecules, the new process actually targets specific molecular fractions, allowing manufacturers to zero in on valuable LAO production. "This means that we are not trying to replace the existing technology," explains Hakim. "We are trying use our process in sync with the original technology so that we can make it more cost effective, while at the same time providing more market flexibility."
Integration with industry is just one form of synchronization on the horizon for the new process; the researchers also emphasize that one of the most exciting possibilities is for their research to create opportunities for collaboration between investigators of biological and chemical conversion technologies. "The idea is that synergy could be developed between biological and chemical routes to LAO production," says Hakim. "For example, biological science researchers could develop microbes—such as E. coli—with metabolic pathways that can process biomass into a form that we can convert to LAOs via chemical reactions."
With all of the complex molecules and long chemical names the process involves, it would be easy to assume that a huge reactor with many parts is required. But Wang points out that another advantage of the process is its simplicity—it is a one-step reaction that can be run continuously in a single, small reactor unit. "Market dynamics are changing all the time, so if I was a plant operator, I would want to install one of these reactor units in my plant so that when market demand for a certain type of LAO increases, I can start making that specific fraction," says Wang.
Before the process can be adopted by industry, further studies and economic analyses must be completed. The researchers are currently studying the process in more detail to better understand their catalyst's properties.
"The most important thing is that our feedstock is renewable, which is a critical advantage in the long run," says Wang, "but this research also has fundamental value for the scientific community in this area. We hope that our work will inspire other researchers in this field to develop improved catalysts to make this process even better."
Journal information: Chemical Communications
Provided by University of Wisconsin-Madison