Using gold and light to study molecules in water
Thanks to a new device that is the size of a human hair, it is now possible to detect molecules in a liquid solution and observe their interactions. This is of major interest for the scientific community, as there is currently no reliable way of examining both the behavior and the chemical structure of molecules in a liquid in real time.
Developed at Boston University by Hatice Altug and her student Ronen Adato, the process brings together infrared detection techniques and gold nanoparticles. It could potentially make a whole new class of measurements possible, which would be a critical step in understanding basic biological functions as well as key aspects of disease progression and treatment. "Our technology could prove useful for studying the behaviour of proteins, medicines and cells in the blood or pollutants in water", says Hatice Altug.
Now a researcher at EPFL Dr. Altug has had her results published in Nature Communications.
Like a guitar string
The device is based on a well-known detection technique called infrared absorption spectroscopy. Infrared light can already be used to detect elements: The beam excites the molecules, which start to vibrate in different ways depending on their size, composition and other properties. "It's like a guitar string vibrating differently depending on its length," explains Hatice Altug. The unique vibration of each type of molecule acts as a signature for that molecule.
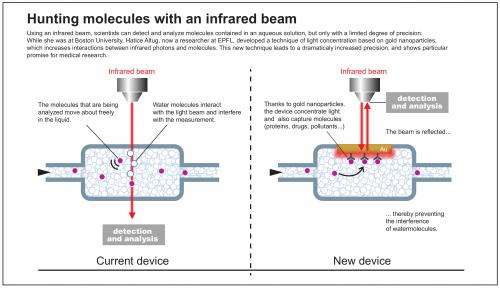
This technique works very well in dry environments but not at all well in aqueous environments. "A large number of molecules need to be present for them to be detected. It's also more difficult to detect molecules in water, as when the beam goes through the solution, the water molecules vibrate as well and interfere with the target molecule's signature," explains Dr. Altug.
Using nanoparticles to capture and illuminate molecules
To get around these obstacles, the researchers have developed a system capable of isolating the target molecules and eliminating interferences.
The size of a penny, the device is made up of miniature fluidic chambers, which are covered on one side with nano-scale gold particles with surprising properties. "We cover the surface of the nanoparticles with, for example, antibodies, in order to make a specific protein or chemical stick to them," explains the researcher. "Once the solution containing the targeted elements is introduced into the chamber, the nanoparticles act as molecule catchers." This technique makes it possible to isolate the target molecules from the rest of the liquid.
But this is not the only role the nanoparticles play. They are also capable of concentrating light in nanometer-size volumes around their surface as a result of plasmonic resonance.
In the chamber, the beam doesn't need to pass through the whole solution. Instead, it is sent straight to the nanoparticle, which concentrates the light. Caught in the trap, the target molecules are the only ones that are so intensely exposed to the photons.
The reaction between the molecules and the infrared photons is extremely strong, which means they can be detected and observed very precisely. "This technique enables us to observe molecules in-situ as they react with elements in their natural environment. This could prove extremely useful for both medicine and biology," states Dr. Altug.
Use in medical research
Another advantage is that the chip is extremely compact and can be connected to microscopes already in use. "We don't need large sample sizes to conduct our analyses," says Ronen Adato.
Going forward, Hatice Altug intends to continue her research with a focus on medical applications. The first tests have been conducted with ordinary antibody molecules, and the analyses now need to be fine-tuned. "I'd really like to work with other life-science researchers, hospitals and biologists. I'm especially interested in using my method in the study of diseases, including cancer and neurological disorders, to observe the effect of certain medicines on diseased cells or to detect disease biomarkers, for example."
More information: www.nature.com/ncomms/2013/130 … /abs/ncomms3154.html
Journal information: Nature Communications
Provided by Ecole Polytechnique Federale de Lausanne