Fast pollutant degradation by nanosheets
(Phys.org) —Waste from textile and paint industries often contains organic dyes such as methylene blue as pollutants. Photocatalysis is an efficient means of reducing such pollution, and molybdenum trioxide (MoO3) catalyzes this degradation. Researchers from Bangalore, India, led by C. N. R. Rao now report no less than four methods to produce nanosheets made of very few layers of MoO3. This material is more efficient as a photocatalyst than bulk MoO3, they write in Chemistry—An Asian Journal.
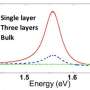
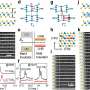
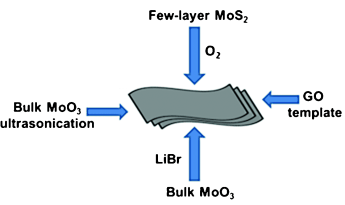
The n-type semiconductor molybdenum trioxide is used widely in heterogeneous catalysis. The Indian team prepared nanosheets of MoO3 by oxidation of MoS2 nanosheets, by using graphene oxide as a template, and by intercalation with LiBr into the bulk material or its ultrasonication. When used as a photocatalyst in the degradation of methylene blue, a heterocyclic aromatic dye, the researchers found few-layered MoO3 to afford nearly complete degradation of the dye in less than 10 minutes, whereas only about one-third of the dye was degraded during this period with the bulk compound.
"As MoO3 holds great potential in applications ranging from gas sensing to energy storage, our study will likely spur further research on few-layer MoO3," says Rao. Indeed, further results reported in their study suggest that a composite of this material with a borocarbonitride is promising as an electrode material for supercapacitors. It will be interesting to see what is coming next for this intriguing few-layer nanostructure.
More information: Rao, C. Synthesis, Characterization, and Properties of Few-Layer MoO3, Chemistry—An Asian Journal. dx.doi.org/10.1002/asia.201300470
Journal information: Chemistry - An Asian Journal
Provided by Wiley