Antioxidant with a long shelf life
(Phys.org) —Scientists from ETH Zurich have developed a nanomaterial that protects other molecules from oxidation. Unlike many such active substances in the past, the ETH-Zurich researchers' antioxidant has a long shelf life, which makes it just the ticket for industrial applications.
There is a lot of talk about antioxidants. These chemical compounds are found in many fruit and vegetable varieties, coffee, tea and red wine, and are generally regarded as healthy. After all, antioxidants protect the body's own proteins and the genetic substance from oxidation. Antioxidants are also used in industry, including as food additives to preserve items for longer or exploit the health aspect as a selling point. They are in food packaging or car tyres to prevent the synthetic material or rubber from becoming brittle. And in the cosmetics industry creams with antioxidants are advertised as anti-aging products.
The problem in using antioxidants, however, is that many of these molecules are not actually very stable in themselves. For instance, they are oxidized in the presence oxygen and gradually lose their antioxidant effect. Researchers under Yiannis Deligiannakis, a visiting professor at the Institute of Process Engineering, have now developed a special nanoantioxidant that is considerably more stable than its conventional counterparts, which means it can be stored more easily and is effective in smaller amounts.
Nanoparticles prevent interaction
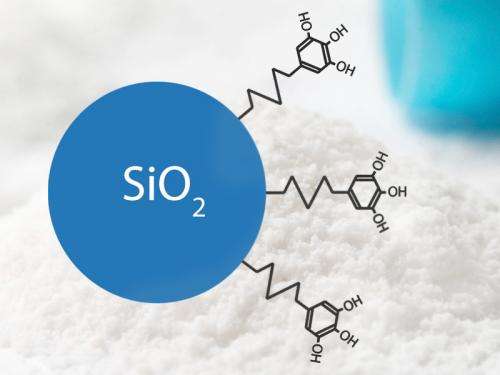
The ETH-Zurich scientists' nanoantioxidant is composed of a silicon dioxide nanoparticle and the naturally occurring antioxidant gallic acid, whereby the two parts are firmly bonded. "Gallic acid is one of the molecules with the best antioxidant activity," explains Georgios Sotiriou, who was involved in the project as a postdoc at the Institute of Process Engineering before moving to Harvard University. However, as with other antioxidants, gallic acid molecules soon lose their effect, especially since they like to latch onto other gallic acid molecules and thus deactivate each other. By combining them with the silicon dioxide, however, the researchers were able to suppress this process. After all, the large nanoparticles compared to the gallic acid molecules prevent the latter from interacting with each other: for reasons of space, they are no more capable of doing so than passengers in two hot-air balloons are of reaching out and touching each other.
"Our nanoantioxidant has the same outstanding effect as gallic acid, but can be sold as a product with a long shelf life for industry," says Deligiannakis. Moreover, the nanoantioxidant is temperature-resistant and could thus protect food that is pasteurised or polymers that are produced at high temperatures. Conventional antioxidants become inactive at these temperatures.
A safe combination
The researchers have now patented their new product and are currently in talks with industrial partners with regard to licensing. The scientists do not expect any major hurdles as far as the safety of the molecule is concerned: both gallic acid and the silicon dioxide nanoparticles are deemed harmless, have been approved by the authorities – including for use in food – and are in active usage. The scientists thus expect tests to confirm that the combination molecule is also safe for cosmetics and food.
More information: Deligiannakis, Y., Sotiriou, G. and Pratsinis, S. Antioxidant and Antiradical SiO2 Nanoparticles Covalently Functionalized with Gallic Acid. ACS Applied Materials & Interfaces, 2012, 4, 6609-6617, doi: 10.1021/am301751s
Journal information: ACS Applied Materials and Interfaces
Provided by ETH Zurich















