November 2, 2012 report
Answer to energy transfer debate could impact areas from photovoltaics to quantum information
(Phys.org)—The transfer of energy between two molecules spaced just nanometers apart plays a key role in many technologies, including photovoltaics, quantum information systems, lighting, and sensors, as well as in biophysics to measure nanometer distances and in photosynthesis. But an open question in this area is what effect, if any, the surrounding photonic environment has on this nanoscale energy transfer. By designing and performing a carefully controlled experiment to answer this question, scientists have settled the debate and found clues to improving the efficiency of many of the technologies that rely on this process.
The scientists, Christian Blum, Niels Zijlstra, Ad Lagendijk, Allard P. Mosk, and Willem L. Vos from the MESA+ Institute for Nanotechnology at the University of Twente in Enschede, The Netherlands (Lagendijk is also with the FOM Institute AMOLF in Amsterdam), along with Martijn Wubs of the Technical University of Denmark in Lyngby and Vinod Subramaniam of the MIRA Institute for Biomedical Engineering and Technical Medicine in Enschede and the MESA+ Institute, have written a paper on the influence of the environment of energy transfer that will be published in an upcoming issue of Physical Review Letters.
The specific type of energy transfer the scientists investigated is called Förster resonance energy transfer (FRET), which is the dominant energy transfer mechanism on the nanoscale. In FRET, a quantum of excitation energy is transferred from one optical emitter (the donor) to another (the acceptor) in nanometer proximity. Scientists know that the Förster transfer rate can be controlled by three criteria: the spectral properties of the optical emitters, the distance between the optical emitters, and the relative orientations of the emitters' dipole moments (a measure corresponding to their electromagnetic properties).
But the role of the environment's photonic properties on Förster transfer has been much less clear. The photonic properties of the environment are characterized by the number of states that can potentially be occupied by a photon, which is referred to as the local density of optical states (LDOS). Scientists know that an environment's LDOS has a definite impact on some molecular processes; for example, a higher LDOS corresponds to a higher spontaneaous emission rate. Using a more familiar analogy, the researchers explain that the question is similar to asking how our surroundings influence our personal lives in a romantic way.
"When you fancy someone, inviting him or her out for dinner is a great idea," Blum told Phys.org. "The romantic environment may help to fall in love. One may wonder if the romantic environment is the reason for falling in love or if it only helps the affection to show. These matters of the heart are notoriously difficult to disentangle and measure."
So far, it's been equally difficult to determine the role of the photonic environment on energy transfer rates. Previous studies have found conflicting evidence: some experiments have found the energy transfer rate has a linear dependence on LDOS (which is also supported by theory), others have found a dependence on the LDOS squared, and still others have found no dependence. This discrepancy likely arises from imprecise control over experimental conditions, such as controlling the distance between molecules, ensuring exact pairing of one donor with one acceptor, and eliminating cross-talk between neighboring pairs of molecules.
In the current study, the researchers could precisely control these conditions by attaching the energy donor and acceptor molecules to opposite ends of a DNA strand. The double-stranded DNA, which is 15 base pairs long, has a high rigidity that keeps the molecules separated by a precisely defined distance of 6.8 nm. To control the influence of the photonic environment, the researchers positioned the molecular pair very close to a metallic mirror, allowing them to control LDOS values.
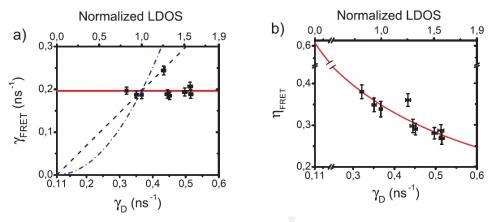
By measuring the Förster transfer of thousands of molecular pairs at various distances from the mirror, the researchers found some surprising results. First, they found that the energy transfer rate is not at all influenced by the distance to the mirror; that is, the environment's LDOS has no effect on the rate.
"It was a great surprise that the LDOS does not influence the energy transfer rate," Blum said. "A previous paper in Science seemed to suggest that the transfer rate would increase linearly with the LDOS. This paper was very influential. Therefore, we originally expected to observe a similar result. At the same time, there was another report saying that the transfer rate would be proportional to the square of the LDOS, which seemed counterintuitive. And, there was work saying that the transfer rate was independent of the LDOS, but unfortunately, the experimental results were not very clear. So it was really confusing when we started."
Nevertheless, Blum said that the new experimental results settle the debate for good.
"We are pleased that our study settles a long-lasting debate," he said. "Our experimental data leaves no room for doubt that the energy transfer rate does not depend on the LDOS; we also performed advanced quantum optical theory that completely confirms this point."
Although the LDOS does not affect energy transfer rate, the results showed that the LDOS does have an impact on the energy transfer efficiency, with a lower LDOS value resulting in a higher efficiency. The researchers showed that, by manipulating the LDOS value, the FRET efficiency could be effectively reduced to zero and increased to 64% in the system studied here. They predict that, by placing a donor molecule that does not undergo any unwanted dark processes in a 3D photonic band gap to shield it from the LDOS, the energy transfer efficiency would approach 100%.
The researchers explained how the efficiency, but not the rate, is impacted by the LDOS.
"From our theory, we have come to realize that the rate of energy transfer depends on the LDOS averaged over an extremely broad distribution of light frequencies, namely, from zero frequency to 10x the typical frequency of the molecules (this corresponds to an extreme ultraviolet wavelength for light)," Blum said. "When averaged over such a broad distribution, the LDOS for most environments of molecules (including our mirrors) does not change at all. Therefore, the energy transfer rate does not depend on the LDOS.
"The reason why the efficiency does depend on LDOS is as follows: the efficiency is the ratio of the transfer rate to the sum of the transfer rate and the light emission rate. So, if the LDOS varies (by changing the distance to the mirror) the transfer rate does not change, but the emission rate does. Therefore the efficiency changes. For example, if the LDOS vanishes, the efficiency is ideally equal to the ratio of transfer rate to transfer rate, which is 100%. While if the LDOS is so large that the emission rate is much greater than the transfer rate, then the efficiency equals the ratio of the transfer rate to emission rate, which tends to 0% efficiency."
The ability to manipulate efficiency by controlling the environment could have implications for the many applications that use FRET. For example, in photovoltaics, FRET is used to transport energy away from the capture site, so increasing the transfer efficiency could increase overall efficiency. In biophysics, LDOS control might improve the way FRET is used to measure molecular distances and interactions. In quantum information applications, a 3D photonic band gap could be used to shield dipole-dipole interactions that store information. Also, in lighting technology, opposite strategies could be used to decrease efficiency since FRET can cause unwanted energy loss.
"Our research does not allow us to draw conclusions about romantic couples," Blum continued the earlier comparison. "But the analogy would be that lovers love each other just as much whether in a romantic environment or not. A romantic environment just makes it more apparent that they have fallen in love. What we wish to illustrate by the analogy is that when the LDOS (in other words, the environment) changes, it does not affect the energy transfer rate between a pair of molecules (in other words, love between two people.) So our analogy is not intended for the transfer efficiency, which is nice in the end, as love is also not about 'efficiency!'"
More information:
Christian Blum, Niels Zijlstra, Ad Lagendijk, Martijn Wubs, Allard Mosk, Vinod Subramaniam and Willem Vos. "Nanophotonic control of the Forster resonance energy transfer efficiency." Physical Review Letters. DOI: 10.1103/PhysRevLett.109.203601
Freely available preprint: arXiv:1207.2272 [physics.optics]
The research groups:
MESA+ Nanobiophysics chair
MESA+ COPS chair
Amolf Photon Scattering group
DTU Fotonik group
Journal information: Physical Review Letters
© 2012 Phys.org