Locating muscle proteins: Scientists bring the basis of muscle movement into sharper focus
Muscle contraction and many other movement processes are controlled by the interplay between myosin and actin filaments. Two further proteins, tropomyosin and troponin, regulate how myosin binds to actin. While theoretical models have in fact described exactly how these muscle proteins interact, this interaction has never previously been observed in detail. Stefan Raunser and Elmar Behrmann from the Max Planck Institute of Molecular Physiology in Dortmund have now managed to image the actin-myosin-tropomyosin complex with an unprecedented accuracy of 0.8 nanometres, which amounts to a resolution of less than one-millionth of a millimetre. This has, for the first time, made it possible to correctly identify the location of proteins within the complex and to analyse the processes involved in muscle contraction. These findings could help determine the impact of genetically determined modifications to the actin-myosin-tropomyosin complex in certain types of hereditary heart disease.
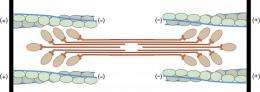
The basic functional unit of a muscle, known as the sarcomere, consists of actin, myosin and tropomyosin proteins. If a muscle is to be able to contract, the myosin must slide along filamentous actin molecules. Working together with troponin, tropomyosin regulates muscle contraction by controlling when myosin binds to actin. In the resting state, tropomyosin and troponin block the binding site for myosin on the actin filament. At this point, the myosin head is at a 90-degree position. Only after an influx of calcium, which docks onto the regulating proteins, is the binding site on the actin filament exposed. The myosin head docks onto this site, changes its conformation and bends in an articulated manner, thereby pulling the actin along with it. As the filaments slide over one another, the sarcomere shortens and the muscle thus contracts.
Scientists from the Max Planck Institute of Molecular Physiology in collaboration with scientists from Hannover Medical School, Ruhr-Universität Bochum and the University of Texas in Houston, have now, for the first time, been able to reveal the details of the interaction on which this model is based. Thanks to improved electron microscopy techniques, Stefan Raunser and his colleagues have also, for the first time, gained an accurate insight into the structural elements of muscle. "This is an important step in understanding the interplay between the individual proteins within the functional structures of muscle", says Raunser.
The scientists were able, for example, to identify the exact location of tropomyosin on the actin filament in the myosin-bound state and, with their detailed imaging of the complex's structure, showed that actin actually brings about conformational changes in myosin. Comparisons with myosin structures in other states have allowed the researchers to describe the interplay of myosin and actin during muscle contraction. "We have, so to speak, drawn a map for biochemists. Our findings will make it easier for them to understand the processes and sequences of events taking place in muscles", explains Raunser.
The findings are also highly relevant from a medical standpoint. The human heart is the body's most important muscle and, if it is working at less than its best, the outcome can be fatal. Malfunctions within the heart are often associated with point mutations. The micrographs taken by the Max Planck researchers have now made it possible to identify the exact location of these mutations for the first time. "Finding the exact location of the mutations is fundamental to developing treatments for such heart diseases", says Stefan Raunser.
More information: Behrmann, Müller, Penczek, Mannherz, Manstein und Raunser, Structure of the Rigor Actin-Tropomyosin-Myosin Complex, Cell, 20th of July 2012
Journal information: Cell
Provided by Max Planck Society