Nanotechnologists develop a 'time bomb' to fight cardiovascular disease
Atherosclerosis, resulting in a narrowing of the arteries and the development of cardiovascular disease, is the leading cause of death worldwide. Until now, no treatment could target diseased areas exclusively, in order to increase drug efficacy and reduce side effects. To help bridge this gap, a group of Swiss researchers from UNIGE, HUG and the University of Basel have developed a veritable 'time bomb,' a treatment that can recognize the diseased areas and treat only them.
In Switzerland, more than 20,000 people (37% of all deaths) die of cardiovascular disease caused by atherosclerosis each year. Treatment options are currently available to people who suffer from the disease but no drug can target solely the diseased areas, often leading to generalized side effects. Intravenous injection of a vasodilator (a substance that dilates blood vessels), such as nitroglycerin, dilates both the diseased vessels and the rest of our arteries. Blood pressure can thus drop, which would limit the desired increased blood flow generated by vasodilatation of diseased vessels and needed for example during a heart attack.
In order to increase the effectiveness of treatments against atherosclerosis and to reduce side effects, a team of researchers from UNIGE, HUG and the University of Basel have developed nanocontainers having the ability to release their vasodilator content exclusively to diseased areas.
Nanotechnology in medicine
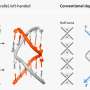
Though no biomarker specific to atherosclerosis has been identified, there is a physical phenomenon inherent to stenosis (the narrowing of blood vessels) known as shear stress. This force results from fluctuations in blood flow induced by the narrowing of the artery and runs parallel to the flow of blood. It is by making use of this phenomenon that the team of researchers has developed a veritable «time bomb», a nanocontainer which, under pressure from the shear stress in stenosed arteries, will release its vasodilator contents.
By rearranging the structure of certain molecules (phospholipids) in classic nanocontainers such as liposome, scientists were able to give them a lenticular shape as opposed to the normal spherical shape. In the form of a lens, the nanocontainer then moves through the healthy arteries without breaking. This new nanocontainer is perfectly stable, except when subjected to the shear stress of stenosed arteries. And that's exactly the intention of this technological advance. The vasodilator content is distributed only to the stenotic arteries, significantly increasing the efficacy of the treatment and reducing side effects. "In brief, we exploited a previously unexplored aspect of an existing technology. This research offers new perspectives in the treatment of patients with cardiovascular disease," explains Andreas Zumbuehl from the Department of Organic Chemistry at UNIGE.
"Nanomedicine is a discipline stemming from general nanoscience but which orients itself towards medical research. The interdisciplinary collaboration between chemistry, physics, basic science and clinical medicine in a highly technical environment could lead to a new era of research," states Till Saxer of the Cardiology and General Internal Medicine Departments at HUG.
"The nano component is present in all disciplines, but the most interesting aspect of nanomedicine is its overview allowing the development of clinical products that integrate this global medical point of view from the earliest onset of research projects," states Bert Müller, Director of the Biomaterials Science Centre (BMC) at Basel.
When chemistry gets involved
How did scientists manage to change the shape of the nanocontainers so that they resemble a lens? By rearranging the structure of molecules, chemists at UNIGE replaced the ester bond that links the two parts of the phospholipid (head and tail), with an amide bond, an organic compound that promotes interaction among phospholipids. Once modified, the molecules are hydrated then heated to form a liquid sphere which will relax to solidify in the form of a lens upon cooling.
The researchers then modelled the cardiovascular system using polymer tubes blocked to varying degrees to represent healthy and stenotic arteries. Next, an artificial extracardiac pump was connected to these arteries in order to reproduce the shear stress induced by the narrowing of the vessels. The nanocontainer was injected into the system and samples were taken from both healthy and stenosed areas. It turns out that the active drug was found in higher concentrations in diseased areas than in non-diseased areas and that the concentrations there were significantly greater than if the drug had been distributed in a homogenous manner.
Provided by University of Geneva