Chemists merge experimentation with theory in understanding of water molecule
(Phys.org) -- Water is the most abundant and one of the most frequently studied substances on Earth, yet its geometry at the molecular level – the simple two hydrogen atoms and one oxygen atom, and how they interact with other molecules, including other water – has remained somewhat of a mystery to chemists.
Most understanding at that level is theoretical, requiring the use of supercomputers to make innumerable calculations over periods of weeks to make educated guesses as to the arrangements and structure of water clusters before they form into liquid water or ice.
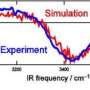
But a new study, using experimentation with a highly advanced spectrometer for molecular rotational spectroscopy, has removed some of the mystery and validates some very complex theory involving the way water molecules bond. It is published in the May 18 issue of the journal Science.
"We set out to determine quantitatively the structure that small assemblies of water adopt, and then compare them to theory to see how well current quantum chemistry predicts the properties of molecules," said Brooks Pate, a chemist in the University of Virginia's College of Arts & Sciences who led the study. "We found experimentally that modern quantum chemistry has reached the point where its theories are proving out in the lab regarding the unusual directional bonding properties of water clusters."
The properties of water, and how it interacts with itself and other molecules, is the basis for many processes in biology, and likely played a major role in the development of life on Earth. But understanding how those bonds form at the molecular level has been largely guesswork.
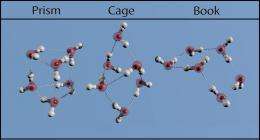
"For the first time, now we have an actual physical picture of what water's molecules put together look like, and it turns out they adopt three different geometries," Pate said. "This is in agreement with theory."
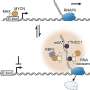
Pate and his U.Va. team identified and imaged a three-dimensional geometry that a water molecule takes on that is the likely precursor structure for forming liquid water and ice.
"We found that the bonding strengths of liquid water actually begin to emerge even in a tiny cluster," Pate said. "The challenge is figuring out how it interacts with other molecules and how the forces between two molecules of water can be described quantitatively, because the orientation of how the waters come at each other makes a big difference in the binding."
There are innumerable possibilities for how this happens, and theorists, including Pate's colleague on the paper, quantum chemist George Shields of Bucknell University, have been working on the details for years without direct validation from experiments.
The difficulty has been in developing techniques that are sensitive enough to image the tiny water molecules and how they orient themselves when interacting with other water molecules. The breakthrough came earlier this year when Pate's U.Va. team used a new tool, a molecular rotational spectrometer developed during the last two years, to make precise measurements that ultimately validated what theory has expressed.
The improved sensitivity of the instrument comes from advances in high-speed digital electronics that provide unprecedented data throughput in the measurement. This core technology is being commercialized for applications in chemical analysis by a Charlottesville start-up venture, BrightSpec.
"This will allow chemists to transfer what we've learned to larger systems," Pate said. "We are checking to see if theory can get right the structures of the arrangements of water molecules so that that information can be used to see how water interacts in larger systems."
The larger systems could include bio-molecules, such as protein in DNA, and how surrounding water molecules might interact with those molecules through hydrogen bonds.
"It is very satisfying to see that the experimental work we did, completely independently of theory, came together so perfectly with the theory," Pate noted.
He said his research is the behind-the-scenes workings of chemistry that ultimately makes up the big picture of how things come together at much larger scales.
"You may not want to know how a bridge was designed, but you sure want to know it was done right," he said. "Likewise, if a theory is used to predict, for example, how a medicine might work, you ideally would want to be able to test the theory to make sure it's right before making the medicine. That would be the ultimate goal – to have theory and experimentation in sync."
Journal information: Science
Provided by University of Virginia