IBN's 'fish and chips' may help accelerate drug discovery

A cheaper, faster and more efficient platform for preclinical drug discovery applications has been invented by scientists at the Institute of Bioengineering and Nanotechnology (IBN), the world’s first bioengineering and nanotechnology research institute. Called ‘Fish and Chips’, the novel multi-channel microfluidic perfusion platform can grow and monitor the development of various tissues and organs inside zebrafish embryos for drug toxicity testing. This research, published recently in Lab on a Chip, has been selected for feature on the journal’s back cover (see Image 1).
Zebrafish, and especially their embryos, are an important model for studying diseases and drug screening. The morphological and molecular basis of tissue and organ development in zebrafish embryos resembles that of humans, and the overall drug toxicity is also comparable with that observed in animals. In contrast to animal models, zebrafish are inexpensive, easily obtainable in large quantity, readily accessible immediately after fertilization, require a shorter development time, and are cheaper to maintain.
Current drug studies on zebrafish embryos are performed on traditional microtiter plates, which do not allow perfusion or the replenishment of growth media and drugs, and cannot facilitate live imaging since the embryos are not fixed in one position due to the size of the well. The conventional way of visualizing tissues and organs in embryos is a laborious process, which includes first mounting the embryos in a viscous medium such as gel, and then manually orienting the embryos using fine needles. The embryos also need to be anesthetized to restrict their motion and a drop of saline needs to be continuously applied to prevent the embryos from drying. These additional precautions could further complicate the drug testing results.
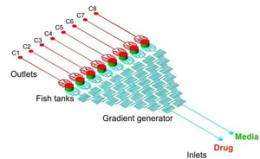
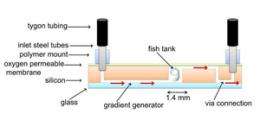
The IBN ‘Fish and Chips’ has been designed for dynamic long-term culturing and live imaging of the zebrafish embryos. The microfluidic platform comprises three parts: 1) a row of eight fish tanks, in which the embryos are placed and covered with an oxygen permeable membrane, 2) a fluidic concentration gradient generator to dispense the growth medium and drugs, and 3) eight output channels for the removal of the waste products (see Image 2). The novelty of the ‘Fish and Chips’ lies in its unique diagonal flow architecture, which allows the embryos to be continually submerged in a uniform and consistent flow of growth medium and drugs (see Image 3), and the attached gradient generator, which can dispense different concentrations of drugs to eight different embryos at the same time for dose-dependent drug studies.
Other key design elements include customizing the dimensions of the fish tank, which is over 200 times smaller than the individual well of a microtiter plate, to fit the embryos exactly in the tank and restrict its movement for live imaging. This is crucial to monitor the growth and development of the various tissues and organs in the embryos. The microfluidic structure was also fabricated using silicon and glass, which provide greater accuracy and reproducibility for commercial application, in comparison with polydimethylsiloxane (PDMS) polymeric-based chips.
Using high-resolution bright-field and fluorescence imaging, the researchers were able to observe the development of various organs such as the eyes, ears, melanophores, brain, yolk sac, trunk and chorion (see video 1), as well as heartbeats (see video 2) in the zebrafish embryos. The researchers also conducted drug toxicity testing on the ‘Fish and Chips’ with valproic acid (VPA), a drug which causes birth defects if consumed by women during their pregnancy. VPA caused abnormality in the development of the eyes and tail in the embryos. The findings clearly established the proof-of-concept that IBN’s ‘Fish and Chips’ could be used as an organ-level drug screening model.
Professor Hanry Yu, IBN Group Leader, who led the research efforts at IBN, said, “Toxicity is a major cause of drug failures in clinical trials and our novel ‘Fish and Chips’ device can be used as the first step in drug screening during the preclinical phase to complement existing animal models and improve toxicity testing. The design of our platform can also be modified to accommodate more zebrafish embryos, as well as the embryos of other animal models. Our next step will involve investigating cardiotoxicity and hepatoxicity on the chip.”
“Miniaturization is being explored in various ways by our researchers to revolutionize drug development and disease diagnosis. This latest microfluidic platform developed by IBN enables researchers to cut down the time and cost of drug testing significantly. Our technology is available for licensing to companies, and we are also open to collaboration to develop customized assays for drug testing,” added Professor Jackie. Y. Ying, IBN Executive Director.
This multi-channel microfluidic perfusion platform was developed in collaboration with Dr. Danny van Noort of the MechanoBiology Institute, Singapore, who provided guidance on chip design and Associate Professor Vladimir Korzh’s group at the Institute of Molecular and Cell Biology, who provided the transgenic zebrafish lines and fish culture expertise.
More information:
D. Choudhury, et al., “Fish and Chips: A Microfluidic Perfusion Platform for Monitoring Zebrafish Development,” Lab on a Chip, 12 (2012) 892-900.
D. Choudhury, et al., “A Microfluidic Device for Studying Drug Toxicity in Developing Zebrafish Embryos,” 6th International Conference on Microtechnologies in Medicine and Biology, 4-6 May 2011, Lucerne, Switzerland.
D. van Noort, et al., “In Vivo Drug Testing in Microfluidics on Medaka Fish Embryo,” 13th International Conference on Miniaturized Systems for Chemistry and Life Sciences, 1-5 November 2009, Jeju, Korea
Journal information: Lab on a Chip
Provided by Agency for Science Technology and Research (A*STAR)















