How nitric acid overcame its fear of water with a little help from its friends
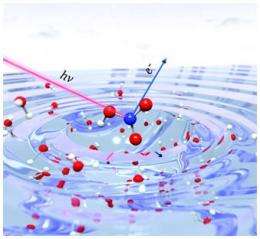
Whether it is a pond or raindrop, when a common nitrogen-containing acid encounters water's surface, it typically falls apart, dissociating into two charged particles. Except, sometimes it is able to hold it together, not entirely but noticeably. Researchers discovered that nitric acid, or HNO3, dissociates less because it gains structure by building more connections at higher concentrations. When enough acid is present, the molecules build hydrogen bonds that reduce dissociation. At lower concentrations, these bonds don't form.
A collaborative team of theorists and experimentalists from Pacific Northwest National Laboratory, University of California at Irvine and Helmholtz-Zentrum Berlin for Materials and Energy, also providing a light source used in the experiments, joined together to obtain these results.
How do you stop smog without halting the economy? That's a question near and dear to the hearts of leaders in the scientific, industrial, and political communities. To find ways to deal with the nitric acid in smog, we must first understand if key reactions involve the acid or its dissociated products. If the reactions involve some of each, how much? Knowing what is happening at the fundamental level gives scientists and other stakeholders the right foundation to move forward. Further, nitric acid is a common prototype that is a good starting point for dissociation studies.
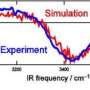
Researchers analyzed the behavior of nitric acid in water. Did it dissociate at the interface or stay together? How about deeper into the water? The team used x-ray photoelectron spectroscopy. The instruments offered data on the concentration of the nitric acid and its dissociation product, nitrate or NO3-. The scientists saw evidence that nitric acid stayed together more at the interface than they expected, even under conditions where it should have readily dissociated.
To determine what was happening during the experiment, Dr. Christopher Mundy and Linus Pauling Postdoctoral Fellow Dr. Marcel Baer at PNNL performed the first principles molecular simulations and together with UC Irvine's Prof. Doug Tobias and postdoctoral fellow Dr. Abe Stern analyzed the results.
"The experiment cannot see the molecules, but the theorists can," said Mundy.
The theory team used large, complex calculations to simulate the nitric acid and water system. They modeled the acid at the surface of water, known as the air/water interface. Further, the team examined the molecules just slightly below the interface and in the bulk water. The size of the calculations demanded time on supercomputers, which was granted by the U.S. Department of Energy's INCITE effort and other resources. With these calculations, the team could see hydrogen bonds forming and breaking.
The team took the results from the simulations and compared them to the experimental results. The simulations explained the different experimental data. "At higher concentrations, nitric acid will pick up a hydrogen bond without dissociating. This correlates quite well to the experimental data," said Baer.
The bonds created a support structure for the nitric acid, reducing dissociation. The results back up other studies that indicate nitric acid dissociates less at the interface then it does in bulk water.
The theoretical chemistry team at Pacific Northwest National Laboratory is continuing to perform highly detailed calculations to get a better view of ions' behavior at interfaces. This work is an important part of the national laboratory's portfolio of research for the U.S. Department of Energy.
More information: T Lewis, et al. 2011. "Does Nitric Acid Dissociate at the Aqueous Solution Surface?" The Journal of Physical Chemistry C 115(43):21183-21190. DOI: 10.1021/jp205842w
T Lewis, et al. 2011. "Dissociation of Strong Acid Revisited: X-ray Photoelectron Spectroscopy and Molecular Dynamics Simulations of HNO3 in Water." The Journal of Physical Chemistry B 115(30):9445-9451. DOI: 10.1021/jp205510q
Provided by Pacific Northwest National Laboratory