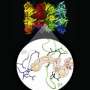
Immunoproteasome crystal structure determined
Like a shredder, the immunoproteasome cuts down proteins into peptides that are subsequently presented on the cellular surface. The immune system can distinguish between self and nonself peptides and selectively kills cells that due to a viral infection present non-self peptides at their surface. In autoimmune diseases this mechanism is deregulated and the immune system also eliminates uninfected cells by mistake. However, inhibition of the immunoproteasome may alleviate disease symptoms and progression. Biochemists at the Technische Universitaet Muenchen (TUM) now succeeded in determining the first crystal structure of an immunoproteasome. The results are reported in the renowned journal Cell and will enable the development of new drugs that selectively target the immunoproteasome.
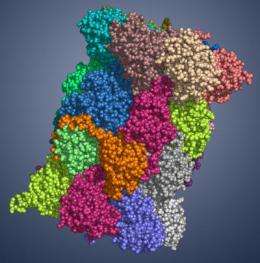
The proteasome, a large, cylindrical protein complex, plays a key cellular role. Like a recycling facility, it breaks down superfluous proteins into smaller fragments that subsequently can be reused. Hence, proteasomes regulate essential cell functions, such as cell survival, growth and division as well as removal of defective proteins.
Apart from the normal, so-called "constitutive" proteasome, mammals also bear a specialized version, the immunoproteasome, which differs from its constitutive counterpart in the enzymatically active subunits. Thus, the immunoproteasome generates more likely peptides that display high affinity for MHC-I receptors. MHC-I molecules carry the peptides generated by proteasomes, the so-called antigens, to the cellular surface and present them to the immune system. Foreign, e.g. viral peptides trigger an immune response, ultimately leading to the elimination of infected cells.
Because of their central role in cell cycle regulation and immune defense, proteasomes represent an excellent drug target; and blockage of both proteasome types already demonstrated therapeutic potential in leukemia. With an annual sale of over 1 billion US $, the first proteasome inhibitor that has been approved for anti-cancer therapy, Bortezomib, is a blockbuster.
As in autoimmune diseases like rheumatism, type 1 diabetes and multiple sclerosis, as well as in certain types of cancer immunoproteasome levels are increased, inhibition of its activity might provide a rational therapeutic strategy. However, so far, the lack of structural data on the immunoproteasome required tremendous efforts to develop selective compounds.
Professor Michael Groll, Chair of Biochemistry in the Department of Chemistry at the TU Muenchen and his co-worker, Eva Maria Huber, in collaboration with Professor Marcus Groettrup, Chair of Immunology at the University of Konstanz (Germany) and director of the Biotechnology Institute Thurgau (BITg) in Kreuzlingen (Switzerland), now achieved a milestone: Using X-ray crystallography, they elucidated the crystal structures of both, the constitutive and the immunoproteasome of mice.
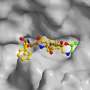
The scientists determined both structures alone and in complex with the compound PR-957 (ONX 0914). PR-957 is a promising proteasome inhibitor that specifically blocks the immuno- but not the constitutive proteasome. However, the molecular reason underlying its selectivity for the immunoproteasome remained unknown. "By visualizing the binding of the inhibitor to both proteasome types on an atomic level, we unraveled the molecular basis for the selectivity of PR-957" explains Groll. "Based on these results, we now can develop new and even more specific inhibitors."
The crystal structures of the constitutive and immunoproteasome enabled Groll and Huber to identify the prerequisites for the high affinity of PR-957 for the immunoproteasome. Remarkably, the orientation of a single amino acid side chain in the substrate binding pocket of the active site is the key to this mystery. Although the substrate binding pockets of both proteasome types are lined with almost identical amino acids, subtle changes in the immunoproteasome cause one particular methionine to adopt a different orientation than in the constitutive proteasome. "This distinct conformation is crucial," emphasizes Eva Maria Huber. "It results in a larger pocket in the immunoproteasome, which therefore preferentially accommodates bulky amino acids and also the inhibitor. In contrast, constitutive proteasomes harbor a significantly smaller cavity that hampers binding of PR-957."
The elucidation of the atomic differences between immuno- and constitutive proteasomes represents an important step for the development of new drugs for the treatment of inflammatory disorders, which often have been linked to elevated levels of immunoproteasomes. The insights gained from the X-ray structures will support the development of new compounds that selectively target the immunoproteasome, without affecting the constitutive counterpart, whose proper function is essential for cellular survival.
More information: Immuno- and constitutive proteasome crystal structures reveal differences in substrate and inhibitor specificity, Eva M. Huber, Michael Basler, Ricarda Schwab, Wolfgang Heinemeyer, Christopher J. Kirk, Marcus Groettrup, Michael Groll, Cell, 17 February 2012
Journal information: Cell
Provided by Technische Universitaet Muenchen