First-of-its-kind study creates new tool for targeted cancer drug development
In a technical tour de force, scientists at Fox Chase Cancer Center have cataloged and cross-indexed the actions of 178 candidate drugs capable of blocking the activity of one or more of 300 enzymes, including enzymes critical for cancer and other diseases. Additionally, a free library of the results has been made available online to the research community. This unique library represents an important new tool for accelerating the development of an entire class of targeted cancer drugs.
The enzymes, called kinases, catalyze a wide array of vital biological activities. Unfortunately, they can also act as drivers for many forms of cancer. For this reason, the candidate drugs, called kinase inhibitors, have the potential to act as powerful anti-cancer agents. They can also interfere with normal processes in the body, however, resulting in side effects. With the new library, researchers will be able to analyze the complex interactions of these inhibitors with their targets to develop cancer drugs that block specific kinases responsible for disease while seeking to avoid major side effects. The results from the Fox Chase team's first-of-its-kind study will appear in the November issue of Nature Biotechnology.
"These results have pushed the field closer to finding truly specific inhibitors of the processes that drive cancer," says Jeffrey R. Peterson, Ph.D., associate professor in the Cancer Biology Program at Fox Chase and senior author on the new study. "We now have a collection of kinase inhibitors that are more well-characterized and understood than any other library. The next step is to use this information to identify specific, effective therapies that stop cancer in its tracks while avoiding healthy processes."
Already, some cancer patients receive kinase inhibitors to treat their disease, and many more such drugs are being developed, says Peterson. But the body contains more than 500 different kinases performing numerous functions. And the vast majority of kinase inhibitors will act on more than one kinase, and so have the potential to interfere with both cancer and the normal processes the body needs for health and survival. Not surprisingly, some of the kinase inhibitors approved for use in cancer cause significant side effects, such as cardiovascular problems.
Until the last few years, however, researchers simply didn't have the technology to observe which kinases a specific inhibitor acted upon. Recently, however, the company Reaction Biology Corporation developed a way to observe the suite of effects from one kinase inhibitor.
For the first time, Peterson and his team catalogued the activity of 178 kinase inhibitors against 300 kinases. The experiment was like observing what happens after shooting a scattergun at a wall of balloons, he says. Before, scientists could only tell if you popped one particular balloon – now, however, they can see if other balloons were hit, as well. "We're essentially shining a light on the wall of balloons so we have a much better view of the balloons that were popped."
Not surprisingly, the researchers found that kinase inhibitors targeted multiple kinases, even some that appeared to be unrelated to each other. They have deposited this massive library of results on a free website so scientists studying kinases and inhibitors can learn more about their multiple interactions.
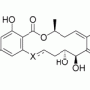
The fact that kinase inhibitors target multiple kinases may actually be a good thing, says Peterson. Initially, scientists had hoped to find an inhibitor that targets one specific kinase involved in cancer; now, they realize that cancer rarely results from one kinase. Instead, multiple kinases likely collaborate to produce the disease -- so to stop that process, you may have to hit all of those kinases together. "It may not be possible to develop a successful drug against one kinase," he says. Indeed, some kinase inhibitors that are effective in cancer -- Sutent (sunitinib) and Sprycel (dasatinib) -- are known to target multiple kinases.
Already, the data have identified inhibitors that act on particular kinases that researchers believe are involved in cancer, but had no known inhibitor – suggesting researchers may one day be able to modify those therapies so they target only those specific kinases and others involved in cancer and avoid kinases unrelated to the disease. In addition, Peterson and his team observed the suite of various kinases affected by cancer drugs that are already in use, with the hope researchers could reduce side effects by modifying the drugs to avoid those healthy kinases.
Provided by Fox Chase Cancer Center