Scientists pinpoint shape-shifting mechanism critical to protein signaling
In a joint study, scientists from the California and Florida campuses of The Scripps Research Institute have shown that changes in a protein's structure can change its signaling function and they have pinpointed the precise regions where those changes take place.
The new findings could help provide a much clearer picture of potential drugs that would be both effective and highly specific in their biological actions.
The study, led by Patrick Griffin of Scripps Florida and Raymond Stevens of Scripps California, was published in a recent edition of the journal Structure.
The new study focuses on the β2-adrenergic receptor, a member of the G protein-coupled receptor family. G protein-coupled receptors convert extracellular stimuli into intracellular signals through various pathways. Approximately one third of currently marketed drugs (including for diabetes and heart disease) target these receptors.
Scientists have known that when specific regions of the receptor are activated by neurotransmitters or hormones, the structural arrangement (conformation) of the receptor is changed along with its function.
"While it's accepted that these receptors adopt multiple conformations and that each conformation triggers a specific type of signaling, the molecular mechanism behind that flexibility has been something of a black box," said Griffin, who is chair of the Scripps Research Department of Molecular Therapeutics and director of the Scripps Florida Translational Research Institute. "Our findings shed significant light to it."
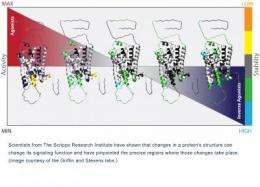
The study describes in structural detail the various regions of the receptor that are involved in the changes brought about by selective ligands (ligands are molecules that bind to proteins to form an active complex), which, like a rheostat, run the gamut among activating the receptor, shutting it down, and reversing its function, as well as producing various states in between.
To achieve the results described in the study, the team used hydrogen-deuterium (HDX) mass spectrometry to measure the impact of interaction of various functionally selective ligands with the β2-adrenergic receptor. A mass spectrometer determines the mass of fragments from the receptor by measuring the mass-to-charge ratio of their ions. HDX has been used to examine changes in the shape of proteins and how these shape changes relate to protein function. The approach is often used to characterize protein-protein interactions that are critical for signal transduction in cells and to study protein-folding pathways that are critical to cell survival.
"At this early stage in understanding GPCR structure and function, it is important to view the entire receptor in combination with probing very specific regions," said Stevens, who is a professor in the Scripps Research Department of Molecular Biology. "Hydrogen-deuterium exchange mass spectrometry has the right timescale and resolution to asked important questions about complete receptor conformations in regards to different pharmacological ligand binding. The HDX data combined with the structural data emerging will really help everyone more fully understand how these receptors work."
"Using the HDX technology we can study the intact receptor upon interaction with ligands and pinpoint regions of the receptor that have undergone change in position or flexibility," Griffin said. "By studying a set of ligands one can start to develop patterns that are tied to activation of the receptor or shutting it down. Once we get a picture of what a functional ligand looks like, it might be possible to develop a drug to produce a highly selective therapeutic effect."
More information: "Ligand-Dependent Perturbation of the Conformational Ensemble for the GPCR b2 Adrenergic Receptor Revealed by HDX," Structure.
Provided by The Scripps Research Institute