Breakthrough in hydrogen fuel cells: Chemists develop way to safely store, extract hydrogen
A team of USC scientists has developed a robust, efficient method of using hydrogen as a fuel source.
Hydrogen makes a great fuel because of it can easily be converted to electricity in a fuel cell and because it is carbon free. The downside of hydrogen is that, because it is a gas, it can only be stored in high pressure or cryogenic tanks.
In a vehicle with a tank full of hydrogen, "if you got into a wreck, you'd have a problem," said Travis Williams, assistant professor of chemistry at the USC Dornsife College.
A possible solution is to store hydrogen in a safe chemical form. Earlier this year, Williams and his team figured out a way to release hydrogen from an innocuous chemical material — a nitrogen-boron complex, ammonia borane — that can be stored as a stable solid.
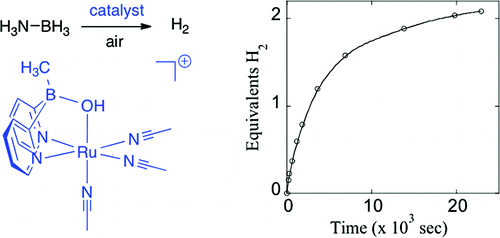
Now the team has developed a catalyst system that releases enough hydrogen from its storage in ammonia borane to make it usable as a fuel source. Moreover, the system is air-stable and re-usable, unlike other systems for hydrogen storage on boron and metal hydrides.
The research was published this month in the Journal of the American Chemical Society.
"Ours is the first game in town for reusable, air stabile ammonia borane dehydrogenation," Williams said, adding that the USC Stevens Institute is in the process of patenting the system.
The system is sufficiently lightweight and efficient to have potential fuel applications ranging from motor-driven cycles to small aircraft, he said.
More information:
A Robust, Air-Stable, Reusable Ruthenium Catalyst for Dehydrogenation of Ammonia Borane, J. Am. Chem. Soc., Article ASAP.
DOI: 10.1021/ja2058154
Abstract
We describe an efficient homogeneous ruthenium catalyst for the dehydrogenation of ammonia borane (AB). This catalyst liberates more than 2 equiv of H2 and up to 4.6 system wt % H2 from concentrated AB suspensions under air. Importantly, this catalyst is robust, delivering several cycles of dehydrogenation at high [AB] without loss of catalytic activity, even with exposure to air and water.
Provided by University of Southern California















