Under pressure, sodium and hydrogen could undergo a metamorphosis, emerging as a superconductor
(PhysOrg.com) -- In the search for superconductors, finding ways to compress hydrogen into a metal has been a point of focus ever since scientists predicted many years ago that electricity would flow, uninhibited, through such a material.
Liquid metallic hydrogen is thought to exist in the high-gravity interiors of Jupiter and Saturn. But so far, on Earth, researchers have been unable to use static compression techniques to squeeze hydrogen under high enough pressures to convert it into a metal. Shock-wave methods have been successful, but as experiments with diamond anvil cells have shown, hydrogen remains an insulator even under pressures equivalent to those found in the Earth's core.
To circumvent the problem, a pair of University at Buffalo chemists has proposed an alternative solution for metallizing hydrogen: Add sodium to hydrogen, they say, and it just might be possible to convert the compound into a superconducting metal under significantly lower pressures.
The research, published June 10 in Physical Review Letters, details the findings of UB Assistant Professor Eva Zurek and UB postdoctoral associate Pio Baettig.
Using an open-source computer program that UB PhD student David Lonie designed, Zurek and Baettig looked for sodium polyhydrides that, under pressure, would be viable superconductor candidates. The program, XtalOpt, is an evolutionary algorithm that incorporates quantum mechanical calculations to determine the most stable geometries or crystal structures of solids.
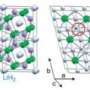
In analyzing the results, Baettig and Zurek found that NaH9, which contains one sodium atom for every nine hydrogen atoms, is predicted to become metallic at an experimentally achievable pressure of about 250 gigapascals -- about 2.5 million times the Earth's standard atmospheric pressure, but less than the pressure at the Earth's core (about 3.5 million atmospheres).
"It is very basic research," says Zurek, a theoretical chemist. "But if one could potentially metallize hydrogen using the addition of sodium, it could ultimately help us better understand superconductors and lead to new approaches to designing a room-temperature superconductor."
By permitting electricity to travel freely, without resistance, such a superconductor could dramatically improve the efficiency of power transmission technologies.
Zurek, who joined UB in 2009, conducted research at Cornell University as a postdoctoral associate under Roald Hoffmann, a Nobel Prize-winning theoretical chemist whose research interests include the behavior of matter under high pressure.
In October 2009, Zurek co-authored a paper with Hoffman and other colleagues in the Proceedings of the National Academy of Sciences predicting that LiH6 -- a compound containing one lithium atom for every six hydrogen atoms -- could form as a stable metal at a pressure of around 1 million atmospheres.
Neither LiH6 and NaH9 exists naturally as stable compounds on Earth, but under high pressures, their structure is predicted to be stable.
"One of the things that I always like to emphasize is that chemistry is very different under high pressures," Zurek says. "Our chemical intuition is based upon our experience at one atmosphere. Under pressure, elements that do not usually combine on the Earth's surface may mix, or mix in different proportions. The insulator iodine becomes a metal, and sodium becomes insulating. Our aim is to use the results of computational experiments in order to help develop a chemical intuition under pressure, and to predict new materials with unusual properties."
Provided by University at Buffalo