Researchers ID molecular link key for cell growth
(PhysOrg.com) -- When a cell is preparing to grow or replicate, it starts the way a monarch planning to expand his territory might: by identifying and marshaling the necessary resources, loading them onto the appropriate vehicles, and transporting them to the front line.
For cells, that means connecting key molecules with so-called motor proteins, which are neatly equipped to recognize them and carry them to their designated positions at the intended site of growth.
In research published Jan. 18 in Developmental Cell, Cornell scientists report on two molecules that work together to initiate that process in yeast cells. The research is a step toward understanding how cells orient themselves within their surroundings and, ultimately, toward finding new ways to fix the process when it goes awry.
In yeast and other organisms, membrane-bound organelles known as secretory vesicles and other compartments are responsible for providing membranes needed to build another cell. In the current research, Anthony Bretscher, professor of molecular biology and genetics, graduate student Felipe Santiago-Tirado and colleagues studied how the motor protein myosin-V, which transports secretory vesicles along filaments of the cytoskeleton, recognizes and binds with its appointed cargo.
They found that two key molecules -- the protein Sec4 and the lipid PI-4P -- work together to facilitate the selection and binding. By requiring two molecular indicators rather than just one, the researches say, cells can closely regulate the cascade of processes that lead to cell growth.
The findings also demonstrate that lipids, normally thought to be just simple barriers, can play a vital role in molecular processes.
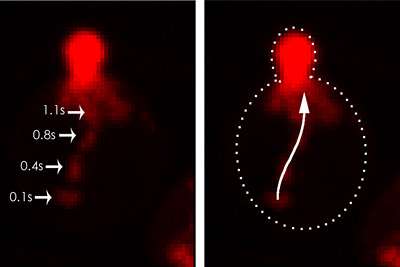
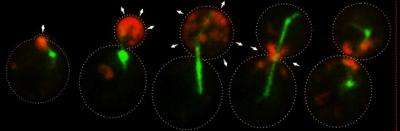
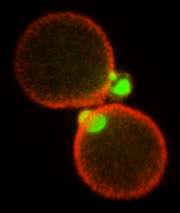
To understand the process, the researchers used confocal microscopy to observe genetically modified yeast cells in which the key protein and lipid molecules were visualized with green fluorescent proteins.
They then tested the cells' ability to grow under a variety of conditions. "By looking at both the lipid and protein markers at the same time, we found that the lipid co-localized with specific proteins and not with others. And it turns out that the ones that co-localized with the lipids are the ones that are associated with secretory compartments," said Santiago-Tirado, who led the study.
If the lipid was present without the protein, or vice versa, the myosin-V protein still trundled along its path to the site of growth -- but with no secretory vesicle attached. "It recognizes these cargoes through both binding a protein, and indirectly through binding a lipid," said Bretscher. "So it's actually a coincidence detector -- it has to bind both."
By greatly boosting either the protein or the lipid when the other element wasn't present, the researchers were able to prompt some cell growth; but the new cells were far less robust.
The research is a step toward deciphering the way cells organize their structure and respond to changes, Bretscher said. The same type of mechanism is likely to apply in mammalian cells, he added.
The work also suggests the need for further studying the role of lipids in cellular processes, Santiago-Tirado said.
"Proteins have been shown to play a role in many things in the cells, but lipids were thought to be just a physical component of the membrane and not involved in this process," he said. "But here we show that lipids also are important -- they do something by themselves."
Ultimately, the findings could lead to treatments for a variety of diseases, Santiago-Tirado said.
"Many diseases are related to the failure of moving these components; so if we understand how the myosin binds to the cargo, then maybe we can try to fix a condition where the myosin is unable to bind to the cargo."
The research was funded by the National Institutes of Health.
Provided by Cornell University