Invention could improve cancer drug delivery, lessen harmful effects of chemotherapy

(PhysOrg.com) -- An invention by University of Arizona researchers may provide a way to deliver chemotherapeutic drugs to cancer tissues in controlled doses without harming healthy body cells.
University of Arizona researchers may have found a way to deliver chemotherapeutic drugs to cancer tissues in controlled doses without harming healthy body cells.
If successful, the invention of gold-coated liposomes could make chemotherapy more effective to destroy cancer cells and alleviate the harmful side effects that can result from the treatment.
The invention by Marek Romanowski, an associate professor in the department of biomedical engineering in the College of Engineering at the UA, and his lab team doesn't have a silver lining. Better: It has a lining of gold. The secret to non-invasively controlling the release of chemotherapeutic drugs lies in nano-scale capsules made of lipids and coated with a fine layer of gold.
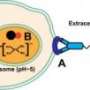
Chemotherapeutic drugs are sometimes encased in small capsules called liposomes, which are made of organic lipids that are already present in human cells. The lipid encasing keeps the body's immune system from attacking the foreign molecule before it can deliver the drug.
Once released into the bloodstream, drug-carrying liposomes accumulate around a cancer tumor because of a property known as leaky vasculature: Tumor cells have extra openings to blood vessels to take in nutrients carried in the bloodstream, usually because they are trying to grow more quickly than normal cells. The extra blood flow means that more nutrients, and also more liposomes, are likely to accumulate in the tumor cells where they eventually break down and release the drug into the cells, leading to cell death.
The highly toxic drugs used for chemotherapy destroy cancer cells, but with no way to discriminate between cell types, they can also damage healthy cells. This damage to the body's normal, healthy cells leads to the side effects normally associated with chemotherapy treatments: anemia, hair loss, vomiting – as cells that make up stomach lining are destroyed – and nausea, among others.
Keys in a lock
To better target cancer cells, the UA team attached liposomes to signal molecules called ligands, which interact with specific cell receptors like keys in a lock.
"It all depends on the disease that we're targeting, but in the case of tumor cells, they over-express certain receptors for several reasons. One is tumor cells are proliferating very quickly, and so they're over-expressing a lot of nutrient receptors because they want to divide faster," said Xenia Kachur, a third-year graduate student in the Biomedical Engineering Graduate Interdisciplinary Program, or GIDP. The extra receptors make the liposomes more likely to latch onto and get inside tumor cells than normal cells.
As they degrade, liposomes release drugs bit-by-bit in an uncontrolled fashion, which may not effectively destroy tumor cells. Said Sarah Leung, a fourth-year graduate student in the biomedical engineering GIDP who also is in the Romanowski lab: "There's a particular concentration at which you have optimal results, so below that you don't have enough of the drug to get a good response, and above that it might be even more toxic."
The new invention could allow doctors to control the amount of drug released at a time, and to release the drug only in the tumor region, thereby protecting healthy cells from damage caused by the drug. This is where the gold lining comes in.
Drugs coated in gold
"A property of gold is that it can convert near infrared light into heat," said Kachur. "By putting gold on the surface of these liposomes, we can then put in a stimulus such as near-infrared light. The gold converts the light into heat, the heat causes the liposome to become leaky, and then whatever's really concentrated inside can diffuse out through the leaky liposome."
"Infrared light penetrates the deepest through the body because it interacts the least with most tissues, and it also prevents a lot of the heating that your body might [otherwise] experience," said Kachur.
The theory goes that the amount of infrared light can be varied to control the amount of drug that is released from the gold-coated liposomes.
"By using more or less light, you can release more or less of the drug and time the responses as well, so when you trigger light, some drug will leak, you can trigger it again and have more drug leak, or you can wait a little while, let the drug disperse, do its thing, then trigger it again. It allows for a lot more freedom with the release process," said Leung. "By having this very triggered response you can hit that therapeutic window."
Despite increased blood-flow to tumor cells and the key-in-lock action of the ligands, some liposomes may still end up inside healthy cells. In that case, the gold-coating could potentially act to prevent release of the toxic drug to the healthy cells.
By selectively shining the infrared light only in the tumor region, doctors could make sure only liposomes in the tumor region are able to release the drug.
"Once you know where the tumors are, you can go ahead and point your light source toward those areas. Whatever else is left will leave the body or may be slowly released, but not to as high or as toxic of levels as it would be if you just injected the drug systemically," said Leung.
The invention has another bonus: "The gold-coated liposome is biodegradable, which is one of the best parts of our system," said Leung. Currently there are no approved chemotherapeutic treatments that allow the gold nanostructures to be eliminated from the body by the body's own mechanisms, said Leung.
Kidneys, the organs that normally filter waste molecules out of the blood, have a limit as to the size of molecule they can filter. "Because of the size it degrades into, our system should be clearable via the kidney, which is really unique," said Leung.
There still are many steps to take to test the invention before it could be used in cancer therapy. But if successful, gold-coated liposomes could provide a method to target chemotherapeutic drugs to cancer cells, non-invasively trigger the drugs' release using infrared light and provide a way for the body naturally to filter the drug from the bloodstream.
One day, cancer patients could potentially receive chemotherapy treatments with confidence that the drugs will effectively destroy cancer cells, and without fear of suffering any harmful side effects.
Provided by University of Arizona