Sorting device for analyzing biological reactions puts the power of a lab in a researcher’s pocket (w/ Video)
Fictional candy maker Willy Wonka called his whimsical device to sort good chocolate eggs from bad, an eggucator. Likewise, by determining what enzymes and compounds to keep and which to discard, scientists are aiming to find their own golden eggs: more potent drugs and cleaner sources of energy.
Toward that end, Harvard researchers and a team of international collaborators demonstrated a new microfluidic sorting device that rapidly analyzes millions of biological reactions. Smaller than an iPod Nano, the device analyzes reactions a 1,000-times faster and uses 10 million-fold less volumes of reagent than conventional state-of-the-art robotic methods.
The scientists anticipate that the invention could reduce screening costs by 1 million-fold and make directed evolution, a means of engineering tailored biological compounds, more commonplace in the lab.
"Our finding is not so much a scientific discovery, but the first demonstration of a new technology," says project leader Jeremy Agresti, a former research associate in the lab of co-author David Weitz, Mallinckrodt Professor of Physics and of Applied Physics in the Harvard School of Engineering and Applied Sciences (SEAS) and Department of Physics. "What limits new areas of research in biology and biotechnology is the ability to assay or to do experiments on many different variables in parallel at once."
The team's technology, first reported in the February 8th online Early Edition of the Proceedings of the National Academy of Sciences, bypasses conventional limitations through the use of drop-based microfluidics, squeezing tiny capsules of liquid through a series of intricate tubes, each narrower than a single human hair.
"Each microscopic drop can trap an individual cell and thus it becomes like a miniature test tube," explains Amy Rowat, a postdoctoral fellow at SEAS. "The drops are coated with a surfactant, or stabilization molecule, that prevents the drops from coalescing with each other and also prevents the contents from sticking to the wall of the drops."
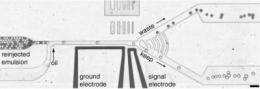
To sort, the system removes inactive and unwanted compounds, dumping the drops into a "bad egg" bin, and guides the others into a "keep" container. Specifically, as the drops flow through the channels they eventually encounter a junction (a two-channel fork). Left alone, the drops will naturally flow towards the path of least fluidic resistance, or the waste channel.
The device identifies the desired drops by using a laser focused on the channel before the fork to read a drop's fluorescence level. The drops with greater intensity of fluorescence (those exhibiting the highest levels of activity) are pulled towards the keep channel by the application of an electrical force, a process known as dielectrophoresis.
"Our concept was to build a miniature laboratory for performing biological experiments quickly and efficiently," explains collaborator Adam Abate, a postdoctoral fellow in applied physics at SEAS. "To do this we needed to construct microfluidic versions of common bench-top tasks, such as isolating cells in a compartment, adding reagents, and sorting the good from the bad. The challenge was to do this with microscopic drops flowing past at thousands per second."
"The sorting process is remarkably efficient and fast. By shrinking down the reaction size to 10 picoliters of volumes, we increased the sorting speed by the same amount," adds Agresti. "In our demonstration with horseradish peroxidase, we evolved and improved an already efficient enzyme by sorting through 100 million variants and choosing the best among them."
In particular, the researchers were struck by the ability to increase the efficiency of an already efficient enzyme to near its theoretical maximum, the diffusion limit, where the enzyme can produce products as quickly as a new substrate can bump into it.
Using conventional means, the sorting process would have taken several years. Such a dramatic reduction of time could be a boon for the burgeoning field of synthetic biology. For example, a biofuels developer could use the device to screen populations of millions of organisms or metabolic pathways to find the most efficient producer of a chemical or fuel. Likewise, scientists could speed up the pace of drug development, determining the best chemical candidate compounds and then evolving them based upon desired properties.
"The high speed of our technique allows us to go through multiple cycles of mutation and screening in a very short time," says Agresti. "This is the way evolution works best. The more generations you can get through, the faster you can make progress."
Provided by Harvard University