Using solar energy to turn raw materials into ingredients for everyday life

(Phys.org) —Just about everything we touch in the course of a day - car, phone, computer, fridge, detergent - even medicines, rely on the chemical industry to turn raw materials such as petroleum by-products, minerals and farm products into valuable chemicals that are the ingredients of life's essential objects.
QUT scientist Dr Sarina Sarina, who achieved outstanding progress in driving this energy intensive chemical production process at ambient temperature using light instead of fossil fuels, has won the prestigious Alexander von Humboldt fellowship at the famous Max Planck Institute in Berlin.
"The problem is it takes a massive amount of electrical energy to make the heat required to convert the crude materials," Dr Sarina, from QUT's Science and Engineering Faculty, said.
"They must be heated to 200 or 300 degrees to achieve the chemical conversion and so this process takes about one third of the energy consumed by manufacturing," Dr Sarina said.
"My research has found that we can achieve high efficiency using sunlight for many chemical productions, and at much lower temperatures.
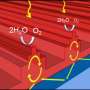
"The key is to use metal nanoparticles such as gold nanoparticles as a 'photocatalyst', which absorbs light and converts the raw materials to useable products.
"All we will need is a giant lens and gold nanoparticles as the photocatalyst to drive the chemical reactions - and all at room temperature so we won't need furnaces."
Dr Sarina said traditional photocatalysts were semi-conductors made of metal oxide with the drawback that they absorbed only UV, not visible light.
"This means traditional photocatalysts use only a small part of the energy available because UV accounts for just 4 per cent of the solar spectrum and 96 per cent of the solar spectrum is visible light and infra-red," she said.
"Previous QUT research by Professor Huai Yong Zhu found that gold nanoparticles absorb most of the solar spectrum so they are very efficient at using visible light.
"Gold nanoparticles, apart from being very expensive, can drive only a small number of reactions but we have found that when we add palladium to the gold nanoparticles it can drive many more reactions than gold by itself.
"I will be working at the Max Planck Institute to develop cheaper, more efficient photocatalysts from bimetallic nanoparticles such as copper or silver and palladium or the other "transition metals" such as iridium and rhodium to find which combination would drive a specific chemical reaction.
"Ultimately, I want to find the perfect photocatalysts to use with Australia's abundant sunshine to convert raw materials using zero fossil fuels for energy."
Provided by Queensland University of Technology