Chemists report promising advance in fuel-cell technology
Creating catalysts that can operate efficiently and last a long time is a big barrier to taking fuel-cell technology from the lab bench to the assembly line. The precious metal platinum has been the choice for many researchers, but platinum has two major downsides: It is expensive, and it breaks down over time in fuel-cell reactions.
In a new study, chemists at Brown University report a promising advance. They have created a unique core and shell nanoparticle that uses far less platinum yet performs more efficiently and lasts longer than commercially available pure-platinum catalysts at the cathode end of fuel-cell reactions.
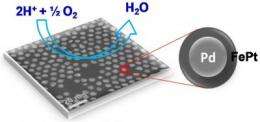
The chemistry known as oxygen reduction reaction takes place at the fuel cell's cathode, creating water as its only waste, rather than the global-warming carbon dioxide produced by internal combustion systems. The cathode is also where up to 40 percent of a fuel cell's efficiency is lost, so "this is a crucial step in making fuel cells a more competitive technology with internal combustion engines and batteries," said Shouheng Sun, professor of chemistry at Brown and co-author of the paper in the Journal of the American Chemical Society.
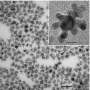
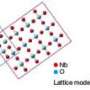
The research team, which includes Brown graduate student and co-author Vismadeb Mazumder and researchers from Oak Ridge National Laboratory in Tennessee, created a five-nanometer palladium (Pd) core and encircled it with a shell consisting of iron and platinum (FePt). The trick, Mazumder said, was in molding a shell that would retain its shape and require the smallest amount of platinum to pull off an efficient reaction. The team created the iron-platinum shell by decomposing iron pentacarbonyl [Fe(CO)5] and reducing platinum acetylacetonate [Pt(acac)2], a technique Sun first reported in a 2000 Science paper. The result was a shell that uses only 30 percent platinum, although the researchers say they expect they will be able to make thinner shells and use even less platinum.
"If we don't use iron pentacarbonyl, then the platinum doesn't form on the (palladium) core," Mazumder said.
The researchers demonstrated for the first time that they could consistently produce the unique core-shell structures. In laboratory tests, the palladium/iron-platinum nanoparticles generated 12 times more current than commercially available pure-platinum catalysts at the same catalyst weight. The output also remained consistent over 10,000 cycles, at least ten times longer than commercially available platinum models that begin to deteriorate after 1,000 cycles.
The team created iron-platinum shells that varied in width from one to three nanometers. In lab tests, the group found the one-nanometer shells performed best.
"This is a very good demonstration that catalysts with a core and a shell can be made readily in half-gram quantities in the lab, they're active, and they last," Mazumder said. "The next step is to scale them up for commercial use, and we are confident we'll be able to do that."
Mazumder and Sun are studying why the palladium core increases the catalytic abilities of iron platinum, although they think it has something to do with the transfer of electrons between the core and shell metals. To that end, they are trying to use a chemically more active metal than palladium as the core to confirm the transfer of electrons in the core-shell arrangement and its importance to the catalyst's function.
Provided by Brown University