September 23, 2009 weblog
Increasing Electric Car Battery Performance

(PhysOrg.com) -- Researchers have found that by replacing conventional graphite electrodes with silicon nanotube electrodes, lithium-ion batteries can store 10 times more charge.
The research was performed at the Stanford University and Hanyang University in Ansan, Korea. The joint effort is between the Universities and LG Chem, a Korean company, who makes lithium ion batteries for the Chevy Volt.
In a conventional Hybrid Car, using today's technology, a lithium-ion battery charge only last 30 minutes.
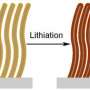
A battery is charged by the movement of lithium ions from the cathode to the anode. By replacing the graphite electrode with silicon, much more energy can be stored because the silicon absorbs larger amounts of lithium in the charging process.
Because silicon anodes absorb up to 10 times more lithium by weight, its volume also increase by 4 times. This can be a disadvantage as the material becomes brittle and can crack after the battery has been charged and discharged a few times. By developing a nanostructured silicon design, researchers have found that the silicon material can better withstand the stress.
There are some major challenges ahead before the silicon nanotube electrodes can be used in electric car batteries. One of the problems is getting back all the energy from the silicon anode after the battery has been charged. The battery must be able to be charged hundreds or thousands of times before this type of battery can be used in electric cars. As of today this has not yet be shown.
Electric car batteries, incorporating silicon nanotube electrodes, could be on the market in three years. It's too early to determine whether this new technology will add to the cost of lithium batteries.
More information: Silicon Nanotube Battery Anodes; Nano Letters, DOI: 10.1021/nl902058c
Via: Technology Review
© 2009 PhysOrg.com