Patent solution in a canning jar

From shopping bags to shampoo bottles to plastic watering cans – many everyday objects both large and small might look very different if it hadn't been for the invention of chemist and Max Planck researcher Karl Ziegler. It took the catalysts developed at the MaxPlanckInstitut für Kohlenforschung (coal research) to pave the way for the use of plastics in everyday items.
Plastics that are currently in widespread use as packaging and various other source materials are made up of polymers. These very large molecules are created when normal-sized molecules join together to form long chains. Indeed, even before Ziegler's catalyst technology, there were processes available to link the components of the molecule chains together, but they were not particularly suitable for mass production. In order to get the polymerization reaction going, the plastics researchers preferred to use, as catalysts, particularly reactive molecules that form free radicals easily.
However, these free radicals are suit able as molecular matchmakers only up to a certain point: they can get the decidedly lethargic precursors really moving, and thus get them lined up relatively quickly, but they have the disadvantage of being difficult to control, and of causing surprising twists when linking the molecule chains.
While they don't form knots and loops, many chemists have, following such experiments with free radicals, found strange polymer chains with undesired branches and other anomalies in their laboratory saucepan. Ziegler's catalysts, on the other hand, would prove to be a reliable and economical alternative for producing the chain molecules.
Together with his coworkers at the Max-Planck-Institut für Kohlenforschung (coal research) in Mülheim an der Ruhr, in 1953, Ziegler discovered how the gaseous olefin ethylene could be converted to poly ethylene relatively easily and, above all, economically. In their lab experiments, they had discovered that mixing aluminum alkyls and titanium tetrachloride initiated the polymerization of ethylene to polyethylene at room temperature – the basis for the first Ziegler catalyst had been found.
Hiking tour with abysses and obstacles
Almost exactly ten years after this discovery, the Director of the Mülheim institute received the Nobel Prize for Chemistry. However, he had to share the award with Italian chemist Giulio Natta, who had developed a similar process for polypropylene based on the work at the Mülheim institute. Ziegler was definitely not thrilled about his co-laureate – and it was most certainly not because he was basically in capable of sharing.
The Nobel Committee in Stockholm was apparently unconcerned about the dis cord between the two chemists. On December 10, 1963, Ziegler and Natta received the Nobel Prize for Chemistry from the hands of King Gustav VI. Adolf of Sweden. In his honorific speech at the Nobel Prize award ceremony, Arne Fredga, a member of the Nobel Committee of the Royal Swedish Academy of Sciences, referred to the particular connection between Ziegler's invention and the interests of the prize founder: "Towards the end of his life, Alfred Nobel was thinking of the manufacture of artificial rubber. Since then, many rubberlike materials have been produced, but only the use of Ziegler catalysts enables us to synthesize a substance that is identical with natural rubber," said Fredga.
Ziegler's and Natta's method for polymerizing polyethylene and polypropylene provided industry with new possibilities for producing easily mouldable plastics in mass quantities.
In his talks, Ziegler himself liked to compare his research path to a "hiking tour through a new country, with ever new and interesting vistas, on which one could frequently overlook a portion of the path to be travelled, but on which one never did know just where the journey was really headed. For decades, I never even remotely thought that my path would also include technological successes.
"At that time, he most likely also never dreamed of the abysses and obstacles he would yet encounter during the course of his journey – and not just scientific ones. In some chapters, the story of one of the most valuable chemistry inventions of all time appears to be an exciting tug-of-war over patents.
Over the span of four decades, the Director of the small Max-Planck-Institut für Kohlenforschung (coal research) fought not only with Giulio Natta, but also with the petrochemical giants for the rights to the new catalysts. It was a battle of David against Goliath, in which, from 1956 to 1999, they had to fend off 149 objections, including patent infringements and legal at tacks on the protective rights. Many contracting partners and a great number of others gave it their all to limit Ziegler's property rights through attempts to prevent the grant of the patents, to restrict their scope, or even to attack their entire legal validity. After all, there was a lot of money involved.
And it had all started out so innocuously in the Mülheim lab. Ziegler wanted to distil alkyl lithium compounds consisting of hydrocarbons and the metal lithium. He had been working on this experiment even before his time in Mülheim. When he attempted to distil ethyl lithium, however, something unexpected happened: in addition to decomposition products of ethyl lithium, the chemists suddenly also found a little 1-butene in the vessel.
FAZ of November 11, 1963
"The Arrangement of Macromolecules on the bestowal of the 1963 Nobel Prize for Chemistry on two plastics scientists
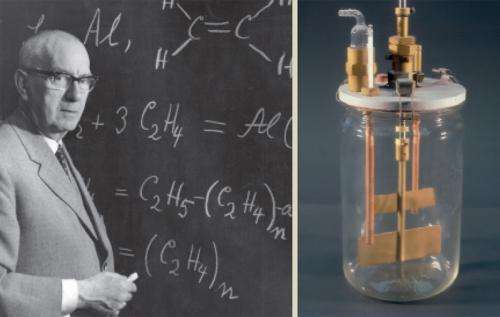
So, during distillation, not only was the ethyl lithium cleaved to form lithium hy dride and ethylene, but apparently there was also an "Aufbau" reaction. Ethyl lithium had added to ethylene – which contains two carbon atoms – to form butyl lithium, which was then cleaved to form lithium hydride and 1-butene with four carbon atoms.
Their curiosity aroused, Ziegler and his staff experimented further and discovered that, when lithium alkyls and excess ethylene are heated up together under pressure, a stepwise synthesis between lithium alkyls and ethylene is possible, and higher lithium alkyls are formed that then decompose in the heat to lithium hydride and α-olefins.
So if, similar to lithium alkyls, lithium hydride could now again be added to ethylene, concluded Ziegler, then it must be possible to produce higher α olefins, or in other words longer hydrocarbons, from ethylene with lithium hydride as a catalyst. When all attempts with the insoluble lithium hydride failed and the research project threatened to hit a dead end, the catalytic reaction of ethylene to α-olefins they had hoped for was finally found with soluble lithium aluminum hydride, which had been discovered just a short time before. Soon, they observed that aluminum hydride likewise adds to ethylene, and the triethyl aluminum that initially forms can, in turn, enter even more efficiently into the stepwise addition to ethylene.
Unexpected effect with revolutionary consequences
What they had hoped for from the com pounds of the rare – and thus expensive – lithium could suddenly be perfectly realized with the widespread and economical aluminum, and further developed for technical applications. In the extreme case, it was possible to link up to 100 ethylene molecules together, but it was not yet possible to produce real polyethylene, like that currently used to make sturdy plastics, with chains composed of 1,000 or more ethylene molecules.
On October 26, 1953, Ziegler's graduate student Heinz Breil undertook the experiment that would revolutionize plastics chemistry. It was the "Aufbau" reaction of triethyl aluminum with ethylene under addition of a zirconium compound. However, the reaction at the usual conditions of 100 degrees Celsius and 100 bar proceeded rather differently than planned, and suddenly the steel autoclave contained a solid white mass of polyethylene.
In the subsequent experiments, it turned out that a similar process was possible with combinations of aluminum alkyls and dialkyl aluminum chlorides with compounds of all transition metals of groups 4, 5 and 6, as well as of thorium, uranium, and under certain conditions, polymerization catalysts can also be produced with other transition metals. The researchers in Mülheim obtained the most effective catalysts with titanium compounds. They owe their discovery that transition metals influence the Aufbau reaction to their own cleanliness in the lab: one colleague had cleaned the reaction vessel with nitric acid. Apparently the acid dissolved nickel out of the vessel wall, causing the reaction to take an unexpected course.
Ziegler's assistant Heinz Martin then managed to take the next important step with experiments in an unusual piece of lab equipment. He polymerized ethylene in a five-liter canning jar from the inventory of Karl Ziegler's wife Maria, using diethyl alu- minum chloride and titanium tetrachloride at normal pressure and room temperature. To do this, he injected the ethylene gas into a stirred suspension of the catalyst in two liters of a suitable solvent. Immediately the temperature rose and, after just a few minutes, flakes of polyethylene formed.
He used cold airflows to keep the canning jar at a temperature of about 70 degrees Celsius. Within about 1.5 hours 400 litres of ethylene gas were polymerized, with the reaction mixture becoming thicker and thicker, until finally, it could no longer be stirred. Depending on the catalyst used, the pasty mass initially appeared grey to brown, but then immediately turned snow-white as soon as it was exposed to air. After washing out the remaining catalyst with alcohol, the chemist obtained about 400 grams of dried polyethylene in powder form.
On November 17, 1953, just three weeks after Heinz Breil's key experiment, Karl Ziegler filed a patent application at the German Patent Office, claiming the process for producing high molecular weight polyethylenes. This method was characterized by bringing ethylene, at pressures of more than 10 bar and temperatures of more than 50 degrees Celsius, into contact with organometallic catalysts composed of a mixture of aluminum trialkyls and compounds of the transition metals titanium, zirconi-um, hafnium, vanadium, niobium, tantalum, chromium, molybdenum or tungsten. He named his co-workers Heinz Martin, Heinz Breil and Erhard Holzkamp as co-inventors.
Four additional applications for further developments of the invention, such as the application of the catalysts also at room temperature and normal pressure, as well as the extension of the catalyst components to include uranium com- pounds, dialkyl aluminum chlorides and alkyl compounds of magnesium and zinc followed a short time later. On August 3, 1954, Karl Ziegler finally expanded the claim relating to the polymerizable olefins from ethylene to include α -olefins such as pro-pylene and 1-butene.
Differing interpretations
A short time later, the Director at the Mülheim institute learned of two Italian patent applications for polypropylene that the chemicals company Montecatini had filed on June 8 and July 27, 1954. These patents named Giulio Natta and Giulio Natta, Piero Pino and Giorgio Mazzanti as inventors. One can certainly imagine his surprise upon discovering this, as he himself had already concluded agreements with the Italian chemicals group relating to the inventions from his catalyst kitchen in Mülheim.
In January 1953, the two partners had also signed an agreement regarding the technological utilization of reactions with organoaluminum compounds, which also included subsequent developments and an exclusive license to certain property rights for Italy. Ziegler had, in early 1954, also forwarded to Montecatini his German patent applications with the information on the new catalysts.
In the accompanying letter, Ziegler wrote: "I expect that we have an under- standing between us that any further expansion of this group of new catalysts shall initially be left completely up to us." Those on the other side of the Alps evidently held a different view, and also the question of whether or not the newly discovered organometallic mixed catalysts fell under the contract provisions was apparently interpreted differently in Ziegler's institute and at Montecatini.
As a consultant to Montecatini, Giulio Natta was very familiar with all the details of the licensing agreement, which helped him get started in the new field very quickly. He had begun, as early as February 1954, to rework the experiments Ziegler de- scribed in the patent applications to polymerize ethylene at normal pressure and, finally, to extend polymerization to, among other things, other olefins than ethylene. In this way, with Ziegler's catalysts com- posed of triethyl aluminum or diethyl aluminum chloride and titanium tetrachloride, Natta produced polymers composed of propylene, 1-butene and styrene.
In the Italian patent from June 8, 1954, Montecatini and Giulio Natta laid claim to the production of polypropylene with catalysts of triethyl aluminum and titanium chloride, as well as the solid, crystalline polypropylene products having a regular structure. With the second patent of July 27, 1954, they expanded the claims to include the production of uniform polymers from olefins with four or more carbon atoms and their crystalline polymer products.
The Italian patent applications that were filed without Ziegler's consent or knowledge led to a complicated patent dispute between the Mülheim-based institute and Montecatini that lasted over decades. The main dispute raged between 1960 and 1983 before the patent office and courts in the US.
In the end, the United States Patent and Trademark Office granted Karl Ziegler's US application legalization of priority from August 3, 1954. Thereafter, Montecatini went before the courts for many years in attempts to get the priority question decided in his favour, and to prevent the grant of a patent for the polymerization of propylene and α-olefins to Karl Ziegler, both without success.
Shared patent brings 32 years of protection
Finally, in 1983, a settlement was reached in which Montecatini withdrew all allega-tions and priority claims and paid damages to Mülheim. The priority dispute with Montecatini and Giulio Natta also continually played a role in the long and tedious court proceedings that Karl Ziegler began in 1966 for patent infringement against some US companies. In 1984, the highest appeals court in Washington D. C. ruled thus: "It was Ziegler and his named co-inventors who invented those catalysts and told Natta about them. It is here immaterial who was the first to use those catalysts to polymerize propylene."
A further decision of the USPTO in Washington proved to be an unexpected blessing for the institute in Mülheim: the division of the application that was required during the examination of one of the first patent applications for the US. The examiner was of the opinion that a pro cess for the polymerization of ethylene and α-olefins and the production of catalysts should be assessed independently. The first patent for the Ziegler catalysts was then granted in 1963, while the second, for the protection of the process, wasn't granted until 1978. Since, until recently, US patent law provided patent protection for 17 years from the date of grant, the Mülheim-based institute thus enjoyed patent rights in the US from 1963 to 1995 for a total of 32 years for the polymerization of propylene.
All complaints from American industry due to double patenting, on the other hand, also failed, all the way up to the highest appeals court in Washington. After all, the examiner had, in the late 1950s, officially required the division of the patent, which was never corrected. In the meantime, though, an amended US patent law had come into effect, so that patent protection is now granted for 20 years from the date of application instead of from the date of patent grant, as is common in the rest of the world. Ziegler's legal dispute over the term of his patent is thus the first and the last of its kind.
Provided by Max Planck Society