June 30, 2014 feature
Venus-flytrap-like gripper could capture individual cells in the human body
(Phys.org) —No two biological cells are exactly the same. Even a small biopsied tumor sample contains cells with large variations in their proliferation rate, potential for metastasis, drug responsiveness, etc. However, because of the large size of the tools used to analyze the cells, data collected from tissue samples is often averaged over a multitude of cells. As such, it may not accurately represent the behavior of individual cells of interest. Since analyzing individual cells is very important for designing effective treatments, researchers are working on ways to capture single cells, and lots of them at once.
In a new study published in Nano Letters, researchers from The Johns Hopkins University in Baltimore, Maryland, and the US Army Research Laboratory in Adelphi, Maryland, have designed and fabricated tiny self-folding grippers that can capture individual cells under in vitro and potentially in vivo environments. The grippers can be mass produced, with perhaps 100 million on a 12-inch wafer, and potentially directed to a specific part of the body to capture specific types of cells. Somewhat like the way in which a Venus flytrap captures its prey, the self-folding grippers enclose their arms around target cells, though without killing them. In experiments, the researchers demonstrated that the grippers can capture mouse fibroblast cells in vitro, as well as red blood cells.
"We believe this is an important step towards a goal of capturing and analyzing single cells within the same device in a high-throughput manner under both in vitro and in vivo conditions," David H. Gracias, Professor at The Johns Hopkins University, told Phys.org.
This gripper is not the first device that can capture individual cells. Currently, a wide range of techniques such as optical and microfluidic traps, flow cytometry (in which a laser is used to suspend cells in a stream of fluid), microwells and even miniature robotic devices are available for in vitro single-cell analysis. However, these techniques face problems such as losing their grip on cells or requiring wires and tethers that restrict mobility, limiting their use.
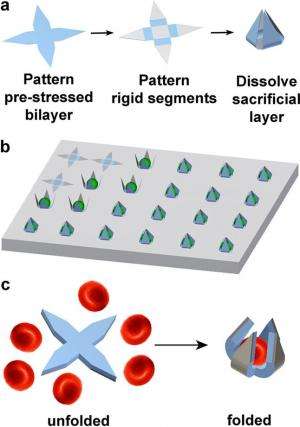
The self-folding gripper developed in the new study overcomes these problems because it has the ability to grip cells using only energy from the release of stress in its own materials, without the need for wires, tethers, or batteries. The gripping mechanism occurs because the gripper's "hinges" are made of a pre-stressed SiO/SiO2 bilayer. The hinges are connected to a rigid body and arms made of only SiO. When exposed to a saline solution, the underlying sacrificial layer releases the arms and causes them to curl upward and close around a cell. As biocompatible and bioresorbable materials, thin films of both SiO and SiO2 dissolve in biological fluids over time.
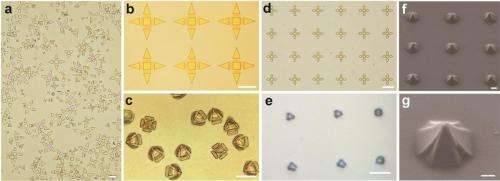
The researchers showed that, using photolithography, the grippers could be fabricated in sizes ranging from 10 to 70 µm from tip to tip when open, which is an appropriate size range to grasp a variety of individual cells. Grippers could be made to fold at angles ranging from 90° to 115° by controlling the bilayer film thickness. Because the grippers have slit openings at the intersection of the arms, nutrients, waste, and other biochemicals can easily flow to and from the cells. Experiments confirmed that the grippers did not kill the cells, although some cells did conform to the shape of the grippers. Because the grippers are optically transparent, they are ideal for imaging the entrapped cells using optical microscopes. Although the timing of the grippers closing cannot currently be controlled, the researchers explain that in the future it may be possible to enable them to respond to and close around specific chemicals.
"Right now the grippers close spontaneously on release from the substrate, so the capture is statistical," Gracias said. "Elsewhere we have shown with larger grippers that a polymer trigger can be added to make such tools responsive to temperature and even enzymes such as proteases. So the single cell grippers could also be potentially made responsive to single cells when coated with the appropriate recognition elements."
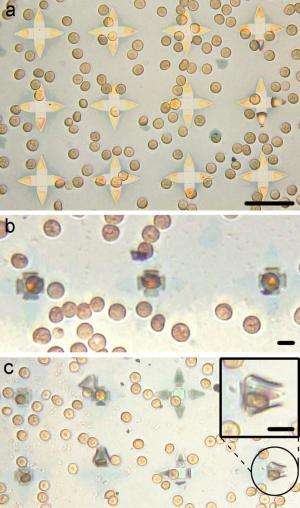
Because the grippers are so small, they have the potential to be used in many parts of the body. For example, they could pass through narrow conduits within the circulatory, central nervous, and urogenital systems. For these in vivo uses, the grippers could be guided by ferromagnetic elements, and patterned biomarkers on them could be used to target specific diseased cells. For in vitro uses, guiding could also be achieved by doping the grippers with magnetic elements such as nickel, and using magnetic fields to move the grippers. Overall, the tiny tools have the potential to forge large improvements in many areas of medicine, which the researchers plan to continue to work on.
"On the in vitro side we are trying to develop a high-throughput assay for capture and analysis of single cells using optical and electrical modalities on a chip," Gracias said. "On the in vivo side, we would like to explore the possibility for biopsy and cell-specific capture in hard-to-reach places in vivo."
More information: Kate Malachowski, et al. "Self-Folding Single Cell Grippers." Nano Letters. DOI: 10.1021/nl500136a
Journal information: Nano Letters
© 2014 Phys.org