June 23, 2014 feature
From barrels to biology: Scientists develop cost-competitive bioderived polymers for a post-petroleum future
(Phys.org) —The advantages of sustainable, biodegradable, carbon-neutral and bioderived renewable polymers – that is, synthetic polymers based on biomolecules produced by living organisms – are reflected in the extent of the research recently conducted into their development. However, such bioderived (or biobased) polymers currently account for a very small percentage of the plastics and elastomers market now dominated by oil-based polymers. (An elastomer, such as rubber, is a polymer that is viscoelastic – that is, both viscous and elastic – with weak intermolecular forces.) To achieve market growth, synthetic polymers have to compete strongly not only in performance, but in large-scale production costs as well – a step that the researchers say is essential for future chemical industry growth. Recently, scientists at the University of Minnesota, Minneapolis, used a biosynthetic approach to combine two factors key to biobased polymers displacing petroleum-based polymers: efficient bioproduction of the branched lactone biomolecule β-methyl-δ-valerolactone (βMδVL) and controlled polymerization. This enabled them to produce economically competitive block polymers (those comprising blocks of different polymerized monomers) based on poly(lactide), or PLA, and poly(βMδVL), or PβMδVL – specifically, PLA-PβMδVL-PLA triblock polymers – having mechanical properties similar to those of today's plastics, including thermoplastic elastomers with properties similar to commercially available styrenic (that is, styrene-based) block polymers.
Prof. Kechun Zhang, Prof. Marc A. Hillmyer, and Prof. Frank S. Bates discussed the paper they and their co-authors published in Proceeding of the National Academy of Sciences. "Since biological feedstocks and petrochemicals are different in their chemical functionalities, it is a significant challenge to generate products from these disparate sources that have comparable properties," Zhang tells Phys.org. "For example, current commercial biodegradable polyesters, including poly(lactide), poly(butylene succinate) and poly(hydroxyalkanoate)s, are brittle, which has limited their broad applications to compete with oil-based polyethylene, acrylonitrile-butadiene-styrene plastic, and other such materials." To address this property barrier, it is necessary to discover new biobased polymers – and since commodity oil-based polymers are priced at around $2–4/kg, a highly-efficient process to produce polymer products from sugar is required to be cost competitive.
At the same time, Zhang points out that there is no reported natural biosynthetic route to βMδVL, and an artificial metabolic pathway has to be designed from the ground up. "To enable biobased production," he explains, "extensive efforts in enzyme discovery, metabolic engineering and bioprocess scale-up are involved. Developing new biochemical products has been an extremely challenging task for the industrial biotechnology sector, with only a few successful examples, such as 1,3-propanediol, isobutanol and 1,4-butanediol, emerging over the past decade."
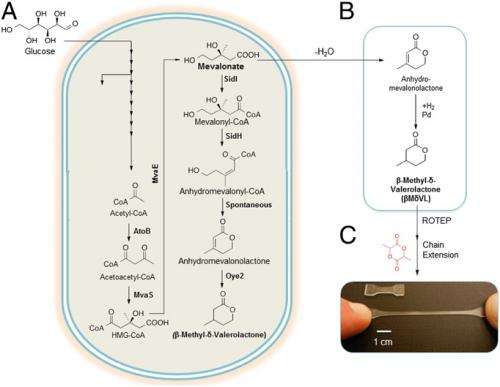
To address the myriad challenges they faced, the scientists employed a synthetic biology approach to develop the first biobased route to βMδVL. "The overall process integrates fermentation, chemical reaction and purification, with a yield suggesting that βMδVL could be potentially produced for less than $2 a kilogram," Hillmyer says. "We discovered that the biobased βMδVL could be converted to a high molar mass rubbery polymer by ring-opening polymerization with a 90% yield. We then we combined poly(βMδVL) and poly(lactide) to generate block copolymers with mechanical properties inaccessible by current commercial aliphatic polyesters." (Aliphatic polymers are hydrocarbons not containing a benzene ring.)
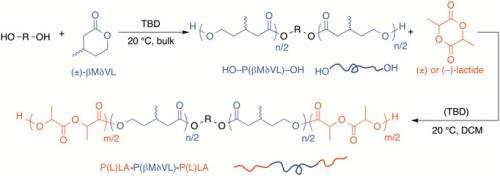
Diving into the details developing a new class of high-performance polyesters through block copolymerization of βMδVL and lactide leading to, as well as of achieving tunable thermal and mechanical properties by controlling molar mass, architecture, and endblock tacticity, Bates says, that poly(βMδVL) has a low glass transition temperature (Tg= –51 °C), making it flexible when temperature is above –51 °C. "On the other hand," he adds, "poly(lactide) has a Tg at 60 °C and is considered a hard polymer at room temperature. We could synthesize various ABA (PLA-P(βMδVL)-PLA) triblock polymers by adjusting the chain length and ratio of the two polymer blocks: high PLA content generates tough plastics, while high P(βMδVL) content generates elastomers."
Relatedly, Hillmyer adds that bioderived polymers have properties similar to commercially available styrenic block polymers. "For example," he illustrates, "with 29% PLA, the ABA triblock polyester behaves much like a rubber band, and can be repeatedly stretched to 18 times its original length without breaking – yet with 59% PLA, the ABA triblock polyester has a property close to that of the material used for tough plastics like high impact polystyrene."
Regarding their creation of a biosynthetic route to produce βMδVL, Zhang explains that the researchers engineered the pathway into the commonly-used fermentation host E. coli. "This artificial pathway shares a common precursor used in cholesterol synthesis – mevalonate – that is converted to anhydromevalonolactone by cloned fungal siderophore proteins. Then the unsaturated lactone is reduced to βMδVL by a reductase from Bacillus subtilis probiotic."
Finally, Hillmyer notes that the use of controlled polymerization techniques to produce well-defined PLA–PβMδVL–PLA triblock polymers was accomplished by adding a solution of lactide directly to a polymerization of βMδVL that was near equilibrium. "Alternatively," he adds, "purfied telechelic poly(βMδVL) could be dissolved in a solution of lactide, and the polymerization initiated by addition of catalyst."
Moving forward, Zhang says that the scientists plan to evolve the artificial pathway to make the total biosynthetic route more efficient – a step, he notes, that will potentially decrease the production of βMδVL to $1.5/kg. In addition, the team will introduce soft poly(βMδVL) to other polymer materials to adjust toughness and biodegradability.
In their paper, the scientists state that their findings establish the groundwork for myriad potential properties and applications. "Based on this first economical biobased elastomer," Zhang says, "we provide unprecedented opportunities for developing sustainable thermoplastic elastomers, agricultural mulch films, chewing gums, pressure sensitive adhesives and new biomedical materials. The application of such biodegradable products could alleviate the environmental impact of current polymer products. In fact," he concludes, "petroleum-based agricultural film waste can render lands uncultivable and becomes a major environmental issue in countries like China; acrylic adhesives create a number of problems for paper recycling; and the cost of chewing gum removal is roughly $2- $3 per piece of gum – and 374 billion pieces of chewing gum are sold worldwide every year."
More information: Scalable production of mechanically tunable block polymers from sugar, Proceedings of the National Academy of Sciences, Published online before print on May 27, 2014, doi:10.1073/pnas.1404596111
Journal information: Proceedings of the National Academy of Sciences
© 2014 Phys.org