Expanding particles to engineer defects: Researchers find that adding an impurity can create order
Materials scientists have long known that introducing defects into three-dimensional materials can improve their mechanical and electronic properties. Now a new Northwestern study finds how defects affect two-dimensional crystalline structures, and the results hold information for designing new materials.
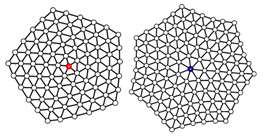
In packed, two-dimensional crystalline systems, such as in photonic two-dimensional crystals, the particles are organized in hexagonal lattices. One particle is in the center of the hexagon with six neighboring particles around it. A defective lattice is when the center particle has one extra or one fewer neighbor, creating a heptagon or pentagon. Two defects of similar types—two pentagons or two heptagons—will repel each other. Two defects of opposite types—one pentagon and one heptagon—will attract one another and proliferate.
"If there is one heptagon or one pentagon, then the structure is strongly distorted," says the paper's coauthor Monica Olvera de la Cruz, Lawyer Taylor Professor of Materials Science and Engineering. "But if you have one pentagon and one heptagon, then the distortion is relieved. The pairs cancel each other out."
Impure particles can cause defects in all types of systems. One impurity is a difference in particle size, which is naturally seen in granular materials, nanoparticles, and colloidal crystals. To see how a size disparity would affect the crystalline order and the system's physical properties, Olvera de la Cruz and postdoctoral fellow Zhenwei Yao devised a model system of soft particles, such as functionalized nanoparticles with grafted chains including nucleic acids or thiols. They made one of the particles in the lattice much larger than the surrounding particles.
"When we expanded one particle, all the neighboring particles were squeezed and stressed," says Yao, coauthor of the paper. "The bigger we made the particle, the more defects it caused."
The larger particle impurity induced defects. Surprisingly, however, instead of repelling one another and distorting the crystalline order, the defects settled into harmony.
"People would expect for them to repel," Olvera de la Cruz says. "But they all came together and arranged to generate a lower energy configuration. The defects around the impurity particles mediate the attractions between impurity particles."
The defects restored order, creating a "screen," or buffer, to protect the rest of the structure from the stress of the added impurity.
This finding could lead to new ways of engineering materials, supporting the Materials Genome Initiative. Creating materials with new properties by adding impurities can be tricky. If the impurities cause defects that induce attractions between impurity particles, then they might create regions where impurities aggregate. "That generates an interface of two materials that can be very damaging," Olvera de la Cruz says. "The impurities have to be very well controlled."
By changing the size of particles, materials researchers may be able to engineer defects in a convenient and precise manner.
More information: The research is described in the paper, "Polydisperity-driven topological defects as order-restoring excitations," published online March 24 in the Proceedings of the National Academy of Sciences. www.pnas.org/content/early/201 … /1403679111.abstract
Journal information: Proceedings of the National Academy of Sciences
Provided by Northwestern University


















