Bacterial immune system has a better memory than expected
Bacteria's memories of hostile viruses are stronger than thought. Even when the intruders change their DNA sequence, the immune system of bacteria can recognise these and subsequently destroy them. That is the conclusion of NWO Vidi researcher Stan Brouns in the journal PNAS.
Bacteria are continuously attacked and killed by intruding viruses. To survive this conflict bacteria have developed an immune system, called CRISPR-Cas (clustered regularly interspaced short palindromic repeats–CRISPR associated). Although there are other less-developed immune mechanisms, the immune system CRISPR-Cas has a 'memory', as a result of which the bacteria are protected against viral infections.
Prior knowledge
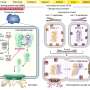
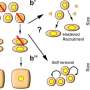
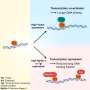
Bacteria add pieces of DNA from intruding phages (small viruses that only infect bacteria) or plasmids (DNA that exchanges information between unicellular organisms) to the memory system of CRISPR-Cas. The DNA of the phages is subsequently broken down and the intruder is rendered harmless. However, phages avoid being recognised by the CRISPR-Cas system by changing their DNA sequence. This makes it harder for the immune system of the bacteria to identify the viral DNA.
Microbiologist Dr Stan Brouns from Wageningen University has discovered that the immune system of bacteria is more robust than had previously been thought: it can recognise viruses by making use of vague memories. Somehow or other the immune system takes up pieces of the now altered DNA and destroys the intruder. In a certain sense the bacterium repeats the same trick but now with prior knowledge. As a vague memory has already been stored in the memory the process now proceeds faster and more efficiently. The CRISPR-Cas system therefore seems able not only to resist recent intruders but also old intruding viruses and intruders related to new viruses.
Yoghurt
The CRISPR-Cas system appears to have a better memory against intruders than had previously been thought; it makes bacteria resistant to external pathogens and therefore gives bacteria an advantage in the evolutionary struggle with intruding viruses. This knowledge can be used to accelerate the resistance of bacteria to phages that threaten milk fermentations (like in yoghurt and cheese).
This research is part of the Vidi project of Stan Brouns. Vidi is an individual grant awarded to talented and creative researchers. The funding instrument makes it possible for researchers to do research of their own choice.
More information: Peter C. Fineran, Matthias J. H. Gerritzen, María Suárez-Diez, Tim Künne, Jos Boekhorst, Sacha A. F. T. van Hijum, Raymond H. J. Staals, and Stan J. J. Brouns. "Degenerate target sites mediate rapid primed CRISPR adaptation." PNAS 2014 ; published ahead of print April 7, 2014, DOI: 10.1073/pnas.1400071111
Journal information: Proceedings of the National Academy of Sciences