October 15, 2013 report
The inheritance of the primary cilium and the soul of the cell
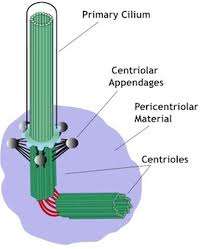
(Phys.org) —The primary cilium plays the role of conductor, and antenna, to many kinds of cells. In photoreceptors, the cilium has been morphed into an expansive pigment-infused photon sieve, while in olfactory cells it is modified to consume the essence of an odorant. Once thought to be captured endosymbionts (as mitochondria are still held to be), cilia are now believed to have been largely eukaryotic inventions rather than the result of prokaryotic acquisitions and mergers. Motile cilia are similar to bacterial flagella, but show several important distinctions. Conventional wisdom holds that when a cell divides, the primary cilium is deconstructed to enable its associated centriole to concentrate on its job as spindle builder. In a paper just released in Cell, researchers now show that a proteinaceous membrane remnant from the hub region of the primary cilium is endocytosed and at mitosis onset, and travels along with the so-called mother centriole as a tiny vesicle. The researchers also show that this lipid attaché is selectively distributed to the favored daughter cell of an asymmetric division, which goes on to retain the stem cell characteristics. This cell is then able to expand its remnant portfolio to get a huge head start over the other daughter cell in building a new cilium.
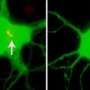
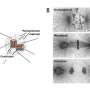
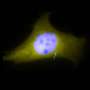
The researchers have dubbed the membrane region in the immediate vicinity of the microtubule core of the cilia, the ciliary membrane (CM). They previously found that a protein marker known found in the CM, known as Arl13b, is closely associated with one of the centrioles in dividing neocortical cells. The Arl13b gene is mutated in a condition known as Joubert syndrome. Short and stubby cilia are one of its characteristic features. By using transmission electron microscopy, the researchers were able to identify Arl13b-positive puncta within vesicles attached to the centriole. The attachment anchors proved to be cilium remnants, and are similar to the fibers that normally link the centriole-derived basal body to the cell membrane during interphase. Basal bodies are known to act as nucleation sites for the growth of cilia microtubules, however the mechanism by which centrioles generate the basal bodies them is still unknown.
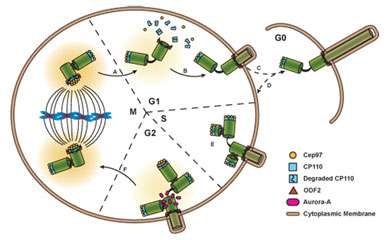
The researchers were able to identify several other components of the CM, including receptors for somatostatin and serotonin. They found that these components also later became closely associated to the centriole. The centriole-CM remnant, and various members of its molecular entourage are linked not only with asymmetry in cell divisions, but appear to act in a larger role as a reliable determinant of cell fate. As the centrioles are duplicated at the onset of S phase in a cycling cell, each daughter centriole remains tightly opposed to the mother. If the mother centriole of a cell has previously built a cilium, the cell might be said to inherit this "memory" for cilia, and produces a new one at a faster rate.
Although it is possible that the two daughter cells remain in communication after division, no kind of "spooky action at a distance" is needed to account for the asynchronous cilia reconstruction. In the developing brain, the primary cilium is needed by cells in order to respond properly to a key diffusable signal known as Sonic Hedgehog. A head start in detecting this or other signals may play an important role in patterning the developing nervous system.
During the early proliferative phase of neurogeneis, cell division is fairly symmetric with both daughters retaining proliferative capacity. Things then progress to a more asymmetric state with one daughter differentiating into a more adult form. Later on, symmetry begins to re-emerge, only now both daughters end up with the adult form. The cilium is now seen as a dynamic organelle that is constantly extended and retracted like the periscope of a submarine, as the cells progress through these stages. Tracing in finer detail its subcellular preservation and status as it weathers mitosis, will help lead to a greater understanding of the soul of the cell.
More information:
Paper: www.cell.com/abstract/S0092-8674(13)01091-X
Commentary: www.cell.com/fulltext/S0092-8674(13)01164-1
Journal information: Cell
© 2013 Phys.org