Tofu-like crystalline catalysts for producing clean energy
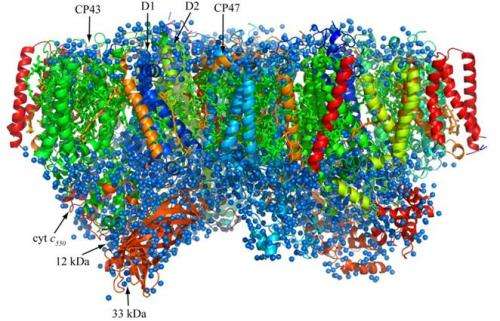
Research by Professor Jian-Ren Shen at Okayama University demystifies the reaction mechanisms of photosynthesis and the findings may lead to the development of methods for producing an unlimited source of clean energy.
Professor Jian-Ren Shen is recognized for his pioneering research on clarifying the fundamental reaction mechanism that governs photosynthetic water splitting, a process with fundamental importance in understanding how oxygenic photosynthetic organisms, such as plants, use energy from sunlight, water, and CO2 to survive.
"I first started research on photosynthetic proteins in the beginning of my doctorate project," says Shen. "Our findings published in 2011 were based on x-ray diffraction experiments of large, high quality single crystal of so-called 'photosystem II' (PS II) at Japan's SPring-8 synchrotron radiation facility at Harima. The ability to produce large sized, single crystals of PS II, an extremely large membrane-protein complex, was critical for determining the crystalline structure of this protein complex to a resolution of 1.9 Angstroms. These results are the culmination of 20 years of my life spent on the development and improvement of the process to produce such large crystals."
Professor Shen's initial research on photosynthesis was focused on clarifying the effects of air pollution on plants. The objectives of this research necessitated clarification of the fundamental mechanism underlying photosynthesis, which in turn required the production of a high quality crystal of PS II. "After many years of exhaustive experiments and uncountable failures, we eventually succeeded in producing large, 'tofu-like' single crystals of PS II with dimensions of 0.7 x 0.4 x 0.1 mm," explains Shen. "This was a major breakthrough that led to the ultra-high resolution analysis of PS II."
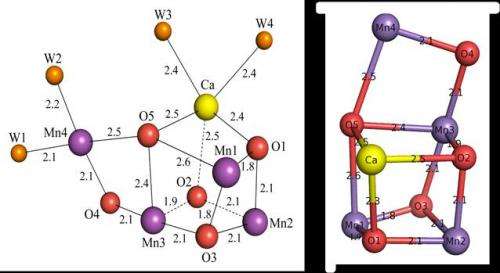
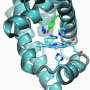
Recent reports on the crystallographic analysis of PS II can be traced back to the early 2000s but the results yielded only 'fuzzy' images because of imperfections in the samples. In contrast the 2011 findings by Shen and colleagues yielded unprecedented images of the core of the PS II protein, showing the existence of cubic-core of four manganese atoms, five oxygen atoms, and a calcium atom, which constitutes the heart of plant life (Science 2011, 334, 1630).
"This cubic structure of Mn4CaO5 acts as a catalyst for the water splitting reaction induced by sunlight," explains Shen. "These results have many important practical applications including the possibility of synthesizing artificial catalyst to dissociate water into oxygen and hydrogen to produce electricity in fuel cells, for example."
Indeed there is increasing interests in 'artificial photosynthesis' for the production of energy. But Professor Shen says that his group will focus on basic research on the reaction mechanism of PS II. "Our next goal is to clarify the so-called 'intermediate structure' of PS II," says Shen. "To do so we require even higher resolution x-ray diffraction experiments at both space and time levels. We are planning to use the SACLA X-ray Free Electron Laser (XFEL) facility in SPring-8 to achieve this. This will enable us to look at the movement of atoms during photosynthesis."
More information:
Yasufumi Umena (1), Keisuke Kawakami (2), Jian-Ren Shen (2) and Nobuo Kamiya (1), Crystal structure of oxygen-evolving photosystem II at a resolution of 1.9 Å; Nature 473, 55–61, (2011).
DOI: 10.1038/nature09913
Provided by Okayama University