Scientists push and pull droplets with graphene
Scientists at the U.S. Naval Research Laboratory (NRL) have moved liquid droplets using long chemical gradients formed on graphene. The change in concentration of either fluorine or oxygen formed using a simple plasma-based process either pushes or pulls droplets of water or nerve agent simulant across the surface. This new achievement offers potential applications ranging from electronics to mechanical resonators to bio/chemical sensors.
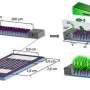
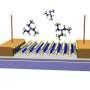
NRL scientists have shown that it is possible to create a chemical gradient on graphene, which pushes or pulls small drops of liquid. Gradients in the wettability of a material are widely found in nature, such as the famous lotus-leaf effect or in spider webs. Researchers who study these effects have found that to be useful, the gradient must be especially smooth without defects that can snag the water droplet. The effect has been achieved before with large molecules or polymers but not with graphene—a layer of carbon only a single atom thick. The chemical flexibility of that carbon enabled both oxygen and fluorine gradients to be created. The mechanical strength of graphene means that these graphene backed chemical gradients could be transferred to many different surfaces. Combined, these advantages provide potential breakthroughs in device design for applications ranging from microfluidics to sensing. The research appears in the June 25, 2013, issue of the journal ACS Nano.
Creating the chemical gradients requires a delicate touch. While graphene is a robust material, it is still only an atom thick—a too-energetic reaction can rip it apart. The ideal solution was to use an NRL-patented plasma processing technology that can produce the necessary wafer-scale chemical patterns when combined with a physical mask. Here, the mask was a canopy that hung over the graphene, but only partially protected it from the plasma. Moving the canopy higher or making it longer creates different gradients, which are clean and smooth without any additional processing steps.
"The beauty of this approach is the ability to rapidly produce chemical gradients of a desired scale or build arrays of multiple gradients over large surface areas. This combination is very desirable when one considers the large-scale fabrication of devices," said Scott Walton, head of the Plasma Applications Section at NRL. "An interesting property of graphene is that it can be transferred to many different substrates," notes co-author Paul Sheehan, of NRL's Chemistry Division. "In principle, one could create this chemical gradient on many different substrates, something which has been hard to date."
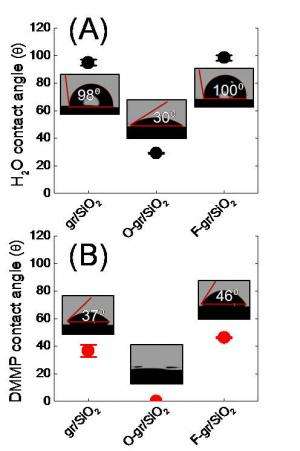
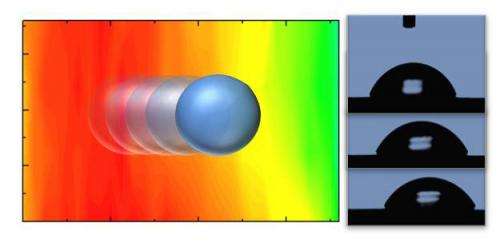
The research team produced and tested two different chemical gradients and then tested them using two liquids, water and dimethyl-methylphosphonate (a nerve agent simulant). For both liquids, a gradient of oxygen functional groups pulled the liquid drops towards increasing oxygen concentration. A fluorine gradient did just the opposite, pushing the droplet towards decreasing fluorine. The direction of motion is broadly attributed to shifts in the surface energy on the functionalized surfaces.
Looking forward, the group believes the chemical gradients could be used to propel smaller droplets and perhaps even single molecules. The ability to move liquids or adsorbates across the surface provides additional capabilities in device design for applications ranging from microfluidics to chemical sensing. "Well-controlled surface modifications provide the ability to manipulate the material attributes locally, individually addressing the sensory and transducing components of a hybrid material, which offer a range of opportunities in a variety of applications," says Sandra Hernandez, the NRC-NRL postdoctoral research associate who designed, fabricated, and characterized the gradients. "You can imagine these films helping to decontaminate a building or clothes by pulling the agent towards an absorber or a catalyst that breaks them down," adds Dr. Sheehan. "Alternately, it could act like a radar dish for a sensor by pulling all the agents in a large area towards a small, low power sensor."
More information: DOI: 10.1021/nn401274e
Journal information: ACS Nano
Provided by Naval Research Laboratory