African Horse Sickness: mapping how a deadly disease might spread in the UK

A disease lethal to horses, until now confined to hot countries, could arrive in the UK. New research creates a picture of its possible spread and pinpoints the area that would be worse hit.
As its name suggests, African Horse Sickness (AHS) is associated with the continent of Africa, where it is feared as a deadly disease. It has long been assumed by British veterinarians and horse-owners that the disease, which is carried by midges, could not spread to cooler northern climates.
But researchers now think that its arrival in northern Europe could be only a matter of time – and perhaps more importantly, that it could spread if it did arrive.
A study undertaken by scientists at the University of Cambridge Department of Veterinary Medicine, in collaboration with the Animal Health Trust and The Pirbright Institute, shows how dangerous it could be for the horse and pony population if AHS was introduced into the UK. The research also identified which regions would be worst hit at different times of the year.
This information could be vital to strategies for coping with an outbreak if it arrived. The study also emphasises the importance of the continued exclusion of the disease.
The research was led by Dr Gianni Lo Iacono, a multidisciplinary scientist whose expertise lies in the mathematical modelling of a range of problems related to the interface between biology and physics. He worked with a team of colleagues from complementary fields including Professor James Wood, a renowned specialist in infectious diseases.
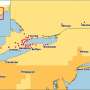
Most strikingly, East Anglia emerges from the study as the region that is most vulnerable to AHS spread which could occur if the disease was not identified early enough for action to be taken to contain it.
In Africa, the disease is spread by infected insects from species of midge known as Culicoides imicola, which carry the African Horse Sickness virus, an orbivirus of the family Reoviridae. Once a horse is infected by AHS, there is no treatment and no cure: the animal will have a high fever within 24 hours and most infected animals will be dead within 48 hours.
Other equidae, zebras and donkeys, are susceptible to AHS infection but do not have such severe disease. Infected zebras do not exhibit any apparent symptoms: as seemingly healthy animals they are potentially lethal carriers. Donkeys develop symptoms but can survive the disease.
First recorded references of AHS occurred in 1327 in Yemen, and in the mid-1600s following the introduction of horses to southern Africa. The disease was clearly identified by the British Army in South Africa 150 years ago when scores of cavalry horses perished in an epidemic.
Ever since, European horse owners have taken comfort from the fact that the disease could not strike in cooler countries. The British climate was considered too cold for the Culicoides imicola midges to survive. On top of this, the UK (and Europe more generally) has protective mechanisms in place that prohibit horses from Africa entering the country.
A growing number of veterinarians now believe that AHS can now arrive in the UK. Well-documented outbreaks were reported in Morocco (1965, 1989–1991), Spain (1987, 1988,1990) and Portugal (1989). The British climate is warming and global transportation of perishable fresh goods – such as flowers and vegetables – offers a possible route for infected midges to enter the country.
The prospect of AHS brings sharply into focus the need for greater research into ways of preventing an incursion of AHS – and ways to cope in the event of an outbreak. "Our work demonstrates that there is no place for complacency about the ability of the virus to spread here," said Professor Wood.
A greater understanding of AHS requires a multi-stranded approach covering the behaviour and life cycle of the midge and the geographical distribution and movement of horses, plus possible routes for infection to enter the country. Midge numbers and activity are highest during the warmer summer months, when the arrival of infection from overseas would be most serious.
In the UK, all horses have passports as a legal requirement but these documents record the owners' address rather than the location where their animals are kept. If horses were mapped according to their owners address, London, for example, would emerge as the centre with the densest horse population. Clearly most horses owned by Londoners are kept outside the city, many of them within easy driving distance of their owners' homes.
Correcting this issue posed problems. However, satellite data on land usage and a survey which recorded the distribution of distances between horses and their owners in different land-use settings (people live closer to their horses in rural areas than they do in urban areas) allowed the researchers to produce a more meaningful map of the risk of the disease. This showed that East Anglia is particularly vulnerable to an outbreak: not only is the region warm and dry, but it also has distinct clusters of horses, notably around Newmarket.
The team has also investigated another important aspect of the disease: the possible 'dilution effect' that could be achieved through keeping animals not susceptible to the virus, such as cattle and sheep, close to horses.
Dr Lo Iacono explained: "In some communities in Africa people keep cattle or sheep near their houses in the belief that this will distract mosquitoes carrying malaria away from people. Some midges show apparent preference for cattle over sheep, so in South Africa deploying cattle to protect sheep from bluetongue (a similar disease to AHS in cattle and sheep) has been proposed as a way to control the disease. On the other hand, the presence of other species might well prove to be an added attraction for midges, exacerbating the threat to horses."
The research re-emphasises the importance of veterinary education to allow early disease identification, which can reduce the critically important reaction times to allow optimal control.
The tools that Dr Lo Iacono has developed have potential applications in mapping and responding to the spread of other diseases, some of which are ecologically even more complex – such as Rift Valley Fever, a mosquito-borne disease that affects both humans and animals, causing a serious disease and in some cases death.
The research provides a good example of how theoretical models can identify biological knowledge gaps (identifying midge biting preferences). This is now being taken forward in other studies.
More information: 'Where are the horses? With the sheep or cows? Uncertain host location, vector-feeding preferences and the risk of African horse sickness transmission in Great Britain' by Giovanni Lo Iacono, Charlotte Robin, Richard Newton, Simon Gubbins, and James Wood is published by the Journal of the Royal Society, Interface (2013) 20130194 doi:10 .1098/rsif.2013.0194
Journal information: Journal of the Royal Society Interface
Provided by University of Cambridge