April 25, 2013 feature
New phase of water could dominate the interiors of Uranus and Neptune
(Phys.org) —While everyone is familiar with water in the liquid, ice, and gas phases, water can also exist in many other phases over a vast range of temperature and pressure conditions. One lesser known phase of water is the superionic phase, which is considered an "ice" but exists somewhere between a solid and a liquid: while the oxygen atoms occupy fixed lattice positions as in a solid, the hydrogen atoms migrate through the lattice as in a fluid. Until now, scientists have thought that there was only one phase of superionic ice, but scientists in a new study have discovered a second phase that is more stable than the original. The new phase of superionic ice could make up a large component of the interiors of giant icy planets such as Uranus and Neptune.
The scientists, Hugh F. Wilson (now at the Commonwealth Scientific and Industrial Research Organisation [CSIRO] in Australia), Michael L. Wong, and Burkhard Militzer at the University of California, Berkeley, have published a paper on the new phase of superionic ice in a recent issue of Physical Review Letters.
Exotic water
As the scientists explain, water has an unusually rich phase diagram, with 15 crystalline phases observed in laboratory experiments and eight additional phases predicted theoretically.
"Superionic water is a fairly exotic sort of substance," Wilson told Phys.org. "The phases of water we're familiar with all consist of water molecules in various arrangements, but superionic water is a non-molecular form of ice, where hydrogen atoms are shared between oxygens. It's somewhere between a solid and a liquid—the hydrogen atoms move around freely like in a liquid, while the oxygens stay rigidly fixed in place. It would probably flow more like a liquid, though, since the planes of oxygen atoms can slide quite freely against one another, lubricated by the hydrogens."
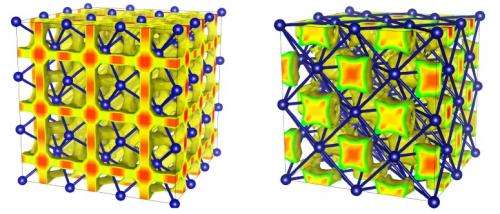
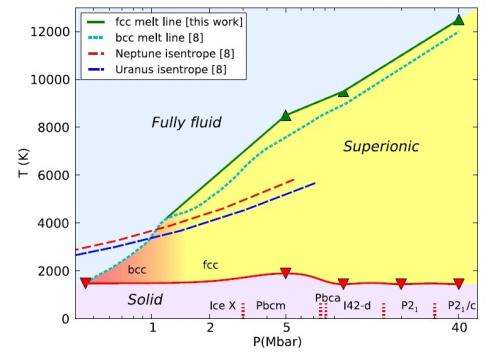
The original phase of superionic ice, called the body centered cubic (bcc) phase, was first predicted with ab initio computer simulations in 1999 by Carlo Cavazzoni, et al. Scientists predict that the bcc phase exists at pressures in excess of 0.5 Mbar (500,000 times greater than atmospheric pressure) and temperatures of a few thousand Kelvin. The bcc phase derives its name from the fact that the oxygen atoms occupy body centered cubic lattice sites. Hints of the bcc phase's instability have been previously observed, but the new study shows for the first time that the bcc phase is less stable than the new phase where the oxygen atoms occupy sites on a face centered cubic (fcc) lattice.
The scientists predict that the fcc phase exists at pressures in excess of 1.0 Mbar, even higher than the pressure for the bcc phase. The scientists' ab initio molecular dynamics simulations also show that the fcc phase has a higher density and lower hydrogen mobility than the bcc phase. That is, the hydrogen atoms in the fcc structure move less frequently to nearby voids between the oxygen atoms, while in the bcc structure, they migrate more freely between different sites. This difference affects the water's thermal and electrical conductivity. In addition, the simulations show that a phase transition between the bcc and fcc phases may exist at pressures of 1.0 ± 0.5 Mbar.
Mysterious planets
Although superionic ice doesn't exist under normal conditions on Earth, the high pressures and temperatures where it is thought to exist are very similar to the predicted conditions in the interiors of Uranus and Neptune.
"Uranus and Neptune are called ice giants because their interiors consist primarily of water, along with ammonia and methane," Wilson said. "Since the pressure and temperature conditions of the predicted new phase just happen to line up with the pressure and temperature conditions of the interiors of these planets, our new fcc superionic phase may very well be the single most prevalent component of these planets."
The researchers predict that understanding superionic ice—particularly the stable fcc phase—will offer insight into these ice giants.
"Uranus and Neptune remain very poorly understood at this stage, and their interiors are deeply mysterious," Wilson said. "The observations we have are very limited—every other planet in the solar system we've visited multiple times, but Uranus and Neptune we've just done brief flybys with Voyager 2. What we do know is that they have bizarre non-axisymmetric non-dipolar magnetic fields, totally unlike any other planet in our solar system. We also know that they're extremely similar in mass, density and composition, yet somehow fundamentally different, because Neptune has a significant internal heat source and Uranus hardly emits any heat at all."
It's possible that the predicted bcc-to-fcc phase transition may explain the planets' unusual magnetic fields, although more research is needed in this area.
"Our results imply that Uranus's and Neptune's interiors are a bit denser and have an electrical conductivity that is slightly reduced compared to previous models," Militzer added.
Understanding Uranus and Neptune's interiors could have implications far beyond our solar system, as well.
"One thing we're learning from the Kepler mission is that Uranus-like or Neptune-like exoplanets are extremely common," Wilson said. "They appear to be more common than Jupiter-like gas giants. So understanding our local ice giants is important, because they're an archetypal example for a huge class of planets out there in the universe."
Future experiments
In the future, the researchers plan to investigate the possible existence of a third superionic phase, as well as attempt to detect the predicted transition between the bcc and fcc phases.
"Shock wave experiments combined with X-ray diffraction techniques will enable one to detect the predicted bcc-to-fcc transition," Militzer said. "Shock waves allow one to reach megabar pressure and heat the sample to thousands of Kelvin at the same time. X-ray diffraction measurements allow one to determine whether the oxygen atoms reside on bcc or fcc positions. This leads to an obvious change in the X-ray diffraction pattern. Shock waves and X-ray diffraction have rarely been combined so far, but Dr. Jon Eggert at Lawrence Livermore National Laboratory is in the process of preparing such experiments."
More information: Hugh F. Wilson, et al. "Superionic to Superionic Phase Change in Water: Consequences for the Interiors of Uranus and Neptune." PRL 110, 151102 (2013). DOI: 10.1103/PhysRevLett.110.151102
Journal information: Physical Review Letters
Copyright 2013 Phys.org
All rights reserved. This material may not be published, broadcast, rewritten or redistributed in whole or part without the express written permission of Phys.org.